Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
* Prices shown are indicative. Clicking "Buy" will take you to the retailer's website. Doctar may earn a small affiliate commission at no extra cost to you.
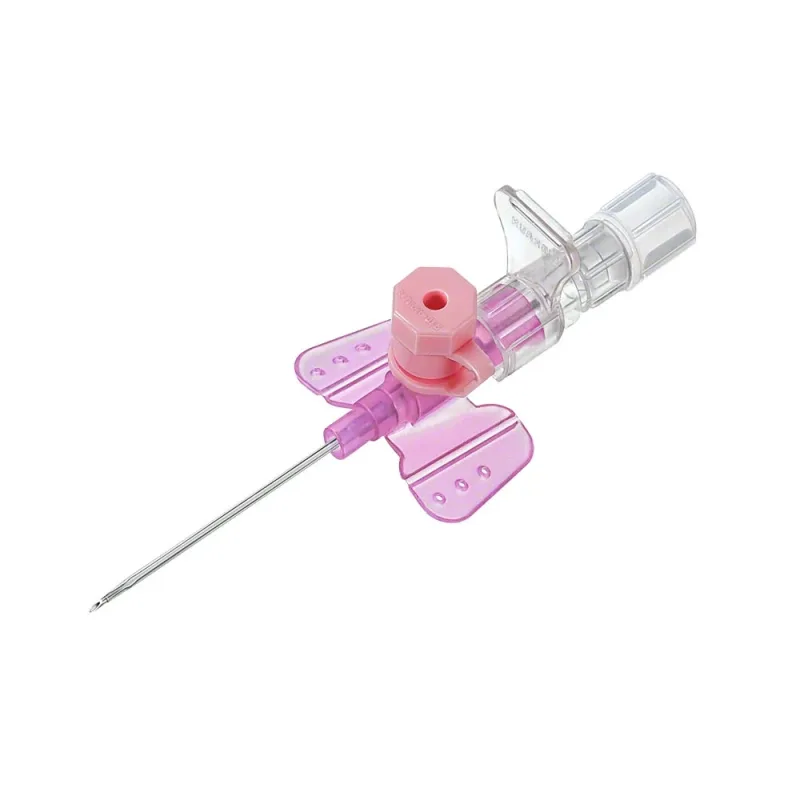
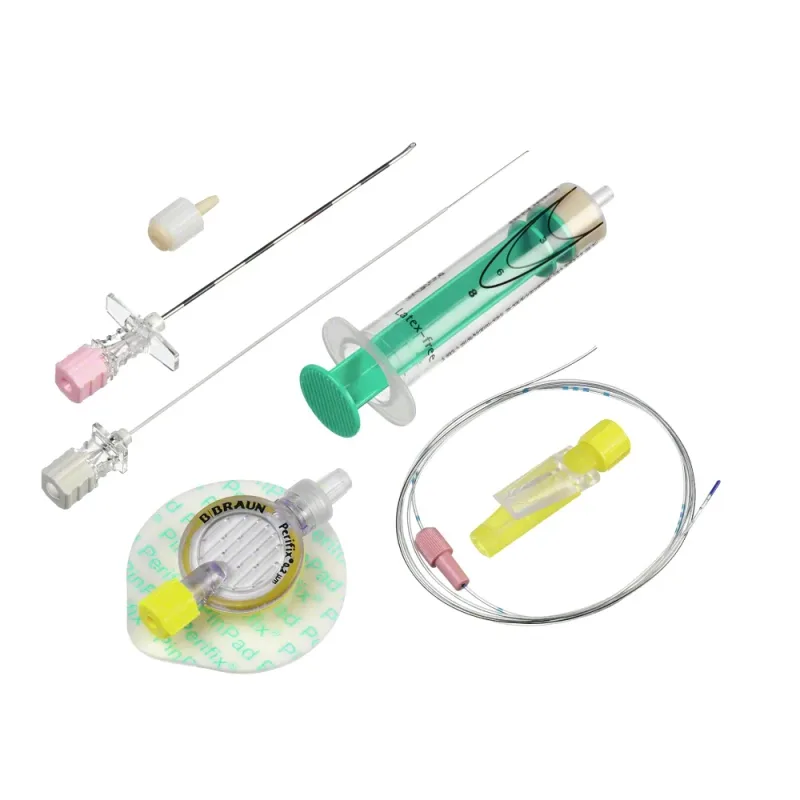
The B. Braun Vasofix Safety IV Catheter with Injection Port is a single-use medical device designed to establish peripheral venous access. It incorporates a passive, automatic safety mechanism intended to prevent needlestick injuries.
The catheter features an integrated injection port, which allows for the administration of medications, fluids, or blood products without the need to disconnect the primary infusion line. This design facilitates efficient and secure delivery of therapies. The device is constructed from materials selected for biocompatibility and patient comfort.
Available in different gauges, it is suitable for a broad range of clinical applications and patient populations, including adults, pediatrics, and neonates. The catheter is DEHP-free, latex-free, and PVC-free, contributing to patient safety by minimizing exposure to certain plasticizers and allergens. Radiopaque markings are incorporated to enable visualization of the catheter under X-ray imaging, aiding in placement verification and monitoring.
The catheter body is available in either PUR (polyurethane) or FEP (fluorinated ethylene propylene) materials, offering clinicians options based on therapeutic needs and patient considerations. Fixation wings are integrated to help stabilize the catheter at the insertion site, potentially reducing movement and associated complications. The design aims to provide a reliable and safe method for intravenous therapy.
The functionality of the Vasofix Safety IV Catheter is based on its integrated safety features and design elements. Upon insertion into a vein, a Double Flashback Technology is employed, which provides confirmation of correct placement by indicating flashback in both the needle hub and the catheter hub. This dual confirmation aims to increase the accuracy of venous cannulation.
The needle features a universal back cut bevel, designed to facilitate smooth insertion into the vein with reduced tissue trauma. Once the catheter is advanced into the vein and the needle is withdrawn, the passive safety mechanism automatically deploys, shielding the needle tip and preventing accidental sharps injuries. The injection port allows for direct access to the catheter lumen for intermittent infusions or flushing.
The radiopaque stripes integrated into the catheter material allow for its detection on X-ray, which is important for confirming placement and for post-procedure monitoring if required. The fixation wings provide a surface for securement to the skin, helping to maintain the catheter's position and minimize dislodgement.
This IV catheter is intended for use by trained medical professionals in various healthcare settings. Its applications span across a wide spectrum of patient care, including the administration of intravenous fluids, medications, chemotherapy, blood products, and for nutritional support. It is suitable for short-term and medium-term venous access.
The availability of different gauge sizes allows for selection based on the intended therapy, patient vein characteristics, and flow rate requirements. For example, smaller gauges may be preferred for pediatric patients or for administering less viscous fluids, while larger gauges might be chosen for rapid infusion of blood products or for higher flow rate therapies. The material options, PUR and FEP, offer different degrees of flexibility and stiffness, which can influence patient comfort and the suitability for different types of infusions.
The primary users of the Vasofix Safety IV Catheter with Injection Port are nurses, physicians, and other healthcare providers responsible for administering intravenous therapies. Patients receiving these therapies are the direct beneficiaries of the device's safety and efficacy. Caregivers may also be involved in monitoring patients with IV access, ensuring the site remains clean and secure.
The device is designed for use in hospitals, clinics, emergency rooms, and other acute care settings, as well as in home healthcare environments where intravenous therapy is managed. Its ease of use and built-in safety features are intended to support efficient workflow and reduce the risk of complications for both the healthcare provider and the patient.
Important usage notes include ensuring the integrity of the packaging before use; the device should not be used if the packaging is damaged. The catheter is designed for single use only and must not be resterilized. Proper aseptic technique should be followed during insertion to prevent infection.
The device should be stored in a cool, dry place, away from direct sunlight, and kept out of reach of children. Following insertion, the catheter site should be regularly monitored for signs of complications such as phlebitis, infiltration, or infection. The specific gauge and material of the catheter should be selected based on the patient's condition, the type of therapy, and the duration of treatment.
The injection port should be flushed according to institutional protocols to maintain catheter patency.

B. Braun
B. Braun Certofix Duo Paed S 408
B. Braun
B. Braun Certofix Duo Central Venous Catheter Kit

B. Braun
B Braun Certofix Trio Central Venous Catheter Kit
B. Braun
B. Braun Certofix Trio Paed S 508

B. Braun
B Braun Combidyn Pressure Monitoring Line, PMO Line
B. Braun
B Braun Ecofix Air Vented IV Infusion Set

B. Braun
B Braun Ecofix Non Vented IV Infusion Set
B. Braun
B Braun Espocan Combined Spinal Epidural Needles