Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
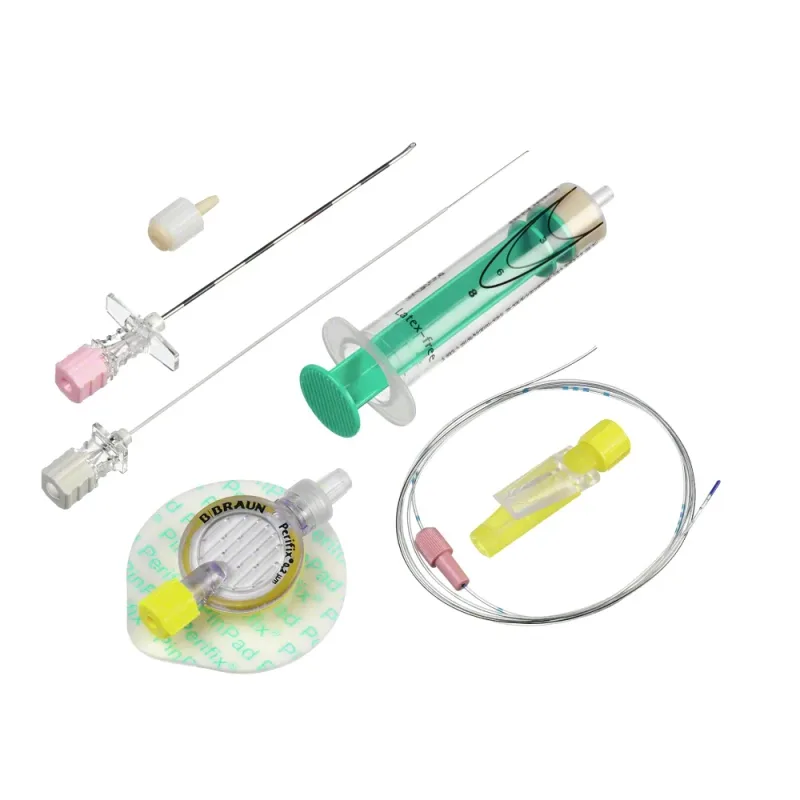
Pack
* Prices shown are indicative. Clicking "Buy" will take you to the retailer's website. Doctar may earn a small affiliate commission at no extra cost to you.
The Espocan Combined Spinal Epidural Needle is a specialized medical device designed for the administration of combined spinal-epidural (CSE) anesthesia. This integrated system allows healthcare professionals to perform both spinal and epidural anesthesia through a single insertion point, streamlining the procedure and potentially reducing patient discomfort. The design facilitates a sequential or simultaneous approach to these regional anesthesia techniques, offering flexibility in clinical practice.
The device is engineered to provide precise needle placement for effective anesthetic delivery in both the subarachnoid and epidural spaces. Its construction aims to ensure the integrity of the spinal and epidural spaces during the procedure. The Espocan needle set typically includes the necessary components for a complete CSE procedure, ensuring that the anesthesia provider has the required tools readily available.
The integration of both needle types within a single unit is intended to simplify the CSE technique.
The mechanism of action for the Espocan Combined Spinal Epidural Needle relies on its dual-needle configuration. The outer needle, a Tuohy needle, is designed for epidural access and features a characteristic curved tip that aids in the safe insertion and guidance of an epidural catheter. This needle also incorporates an additional back eye, positioned within the Tuohy curve, which can be utilized for specific procedural steps.
Inside the Tuohy needle, a specialized spinal needle is housed. This inner spinal needle is designed with a tip that exits through the back eye of the Tuohy needle, allowing for precise positioning within the epidural space before advancing to the subarachnoid space. A sleeve is often incorporated to center the spinal needle within the epidural needle, ensuring stable and controlled advancement.
The spinal needle itself is typically a pencil-point or Quincke-point design, chosen for its ability to separate dural fibers rather than cut them, potentially reducing the incidence of post-dural puncture headache.
Key features of the Espocan Combined Spinal Epidural Needle include its integrated design, which combines a Tuohy needle for epidural access with a dedicated spinal needle for subarachnoid access. The Tuohy needle component is designed with a specific bevel and an additional back eye to facilitate epidural catheter placement and other procedural maneuvers. The spinal needle component is designed for precise entry into the subarachnoid space.
The needles are manufactured with specific dimensions, such as a diameter of 1.30 mm (18 gauge) and a length of 88 mm (3 1/2 inches), to suit various patient anatomies and procedural requirements. Depth markings on the needle shaft may be present to assist the clinician in monitoring insertion depth.
The set typically includes a spinal syringe for aspiration and injection of cerebrospinal fluid and anesthetic agents.
This device is intended for use by qualified medical professionals, specifically anesthesiologists and other physicians trained in regional anesthesia techniques. It is utilized in hospital settings, surgical centers, and labor and delivery suites where regional anesthesia is indicated. Patients undergoing surgical procedures or requiring pain management during labor and delivery may receive CSE anesthesia administered with this device.
Caregivers may be involved in patient preparation and monitoring during and after the procedure. Typical applications include providing anesthesia for lower abdominal surgeries, orthopedic procedures of the lower extremities, and labor analgesia. The combined approach allows for rapid onset of analgesia and anesthesia from the spinal component, while the epidural component provides for prolonged anesthesia or analgesia and the ability to titrate medication as needed.
Important usage notes include the requirement for sterile technique throughout the procedure to prevent infection. The anesthesia provider must be proficient in both spinal and epidural techniques and understand the specific design and intended use of the combined needle. Careful consideration of patient anatomy, contraindications, and potential complications is essential.
The depth markings, if present, should be used as a guide, but clinical judgment remains paramount. Proper handling and disposal of the device after use are crucial to prevent needlestick injuries and ensure biohazard waste management. The specific components of the CSE set, including the spinal syringe and the type of spinal needle (Pencan or Spinocan), are designed to work in conjunction with the Tuohy needle for optimal performance.

B. Braun
B. Braun Certofix Duo Paed S 408
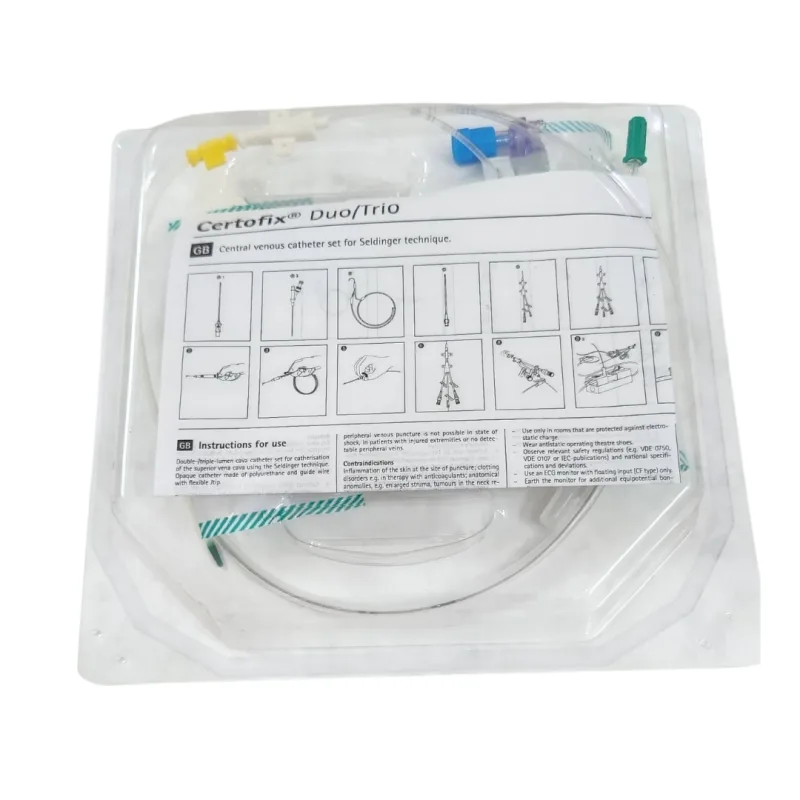
B. Braun
B. Braun Certofix Duo Central Venous Catheter Kit

B. Braun
B Braun Certofix Trio Central Venous Catheter Kit
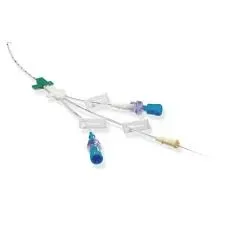
B. Braun
B. Braun Certofix Trio Paed S 508

B. Braun
B Braun Combidyn Pressure Monitoring Line, PMO Line
B. Braun
B Braun Ecofix Air Vented IV Infusion Set

B. Braun
B Braun Ecofix Non Vented IV Infusion Set
B. Braun
B Braun Introcan IV Catheter Cannula