Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.

Pack
* Prices shown are indicative. Clicking "Buy" will take you to the retailer's website. Doctar may earn a small affiliate commission at no extra cost to you.
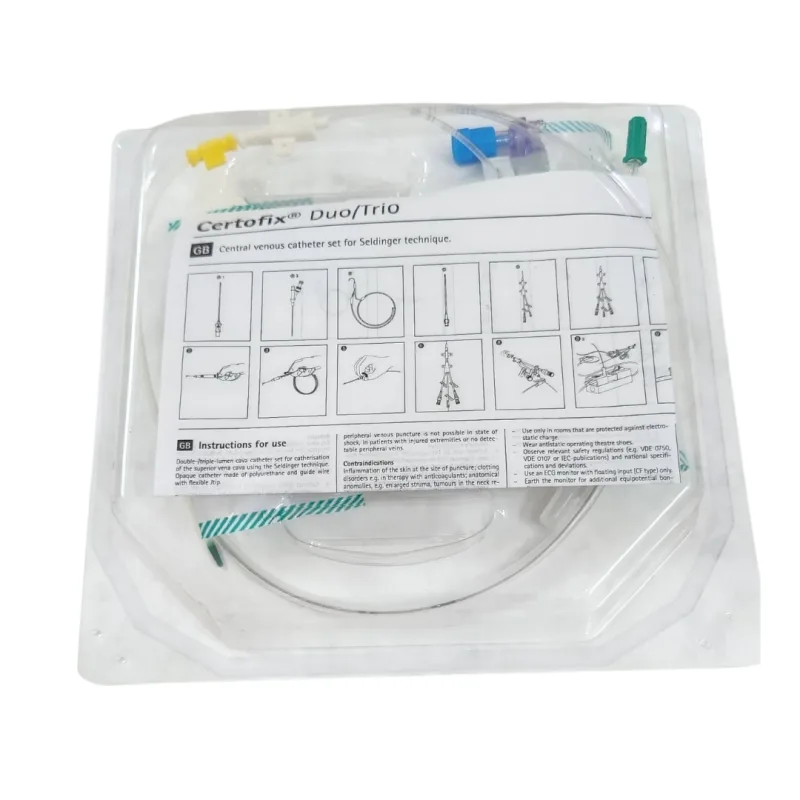
This is a central venous catheter kit designed for the insertion of a triple-lumen catheter into the vena cava. The kit facilitates catheterization utilizing the Seldinger technique, a standard medical procedure for accessing blood vessels. The primary function of this device is to provide reliable and safe access to the central venous system for various therapeutic and diagnostic purposes.
The catheter itself is constructed from a medical-grade material that is detectable by X-ray, allowing for verification of its position within the body after insertion. The transparent tubing allows for visual inspection of blood flow and potential air bubbles.
The mechanism of action involves the use of a guidewire to initially access the target vein. The guidewire, featuring a kink-resistant design and a J-tip for ease of navigation, is advanced into the vessel. A dilator is then used to create a tract for the catheter.
The triple-lumen catheter is subsequently introduced over the guidewire. The presence of three distinct lumens within a single catheter allows for simultaneous administration of fluids or medications, withdrawal of blood samples, and monitoring of central venous pressure, thereby increasing procedural efficiency and patient throughput. The catheter tip is designed to be soft to minimize potential injury to the vascular endothelium during insertion and while in situ.
Key features of this catheter kit include its triple-lumen configuration, which offers versatility in patient management. The material's X-ray detectability is crucial for confirming accurate placement of the catheter tip, a critical step in preventing complications. The kit incorporates a kink-proof guidewire with a flexible J-tip to facilitate smooth insertion and reduce the risk of the wire becoming deformed.
A scalpel is provided for making the initial skin incision, and a dilator is included to prepare the vessel tract. The Safsite connector is designed to maintain a closed system, reducing the risk of air embolism and contamination. Color-coded Luer-Lock hubs on the catheter lumens aid in the unambiguous identification of each port, preventing medication errors or misconnections.
Fixation wings are integrated into the catheter design to secure its position at the insertion site, enhancing stability and reducing the likelihood of dislodgement.
This central venous catheter kit is intended for use by trained medical professionals in various healthcare settings. This includes, but is not limited to, intensive care units, operating rooms, emergency departments, and other areas where continuous or frequent central venous access is required. Patients requiring long-term intravenous therapy, hemodynamic monitoring, or administration of vesicant medications are potential recipients of this type of device.
Caregivers may also be involved in the management of patients with indwelling central venous catheters, following established protocols.
Typical applications for this device include the administration of parenteral nutrition, chemotherapy, antibiotics, and other intravenous medications. It is also used for rapid infusion of fluids, central venous pressure monitoring, and obtaining blood samples for laboratory analysis without the need for repeated peripheral venipuncture. The triple-lumen capability allows for concurrent administration of incompatible medications or simultaneous infusion and withdrawal.
The ECG-based catheter tip placement control feature, when utilized, aids in positioning the catheter tip in the superior vena cava or at the cavoatrial junction, which is considered optimal for central venous access and can help reduce the risk of cardiac arrhythmias or malposition.
Important usage notes include ensuring the integrity of the packaging before use; the product is intended for single use and should not be resterilized. It is essential to follow aseptic techniques throughout the insertion procedure to minimize the risk of infection. The device should be stored in a cool, dry environment away from direct sunlight.
Proper fixation and regular assessment of the insertion site are necessary to maintain catheter patency and prevent complications such as infection, thrombosis, or dislodgement. The ECG monitoring cable, if used, should be connected correctly to the patient's ECG monitoring system and the catheter hub as per the manufacturer's instructions.

B. Braun
B. Braun Certofix Duo Paed S 408
B. Braun
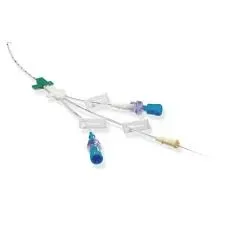
B. Braun Certofix Duo Central Venous Catheter Kit
B. Braun
B. Braun Certofix Trio Paed S 508

B. Braun
B Braun Combidyn Pressure Monitoring Line, PMO Line
B. Braun
B Braun Ecofix Air Vented IV Infusion Set

B. Braun
B Braun Ecofix Non Vented IV Infusion Set
B. Braun
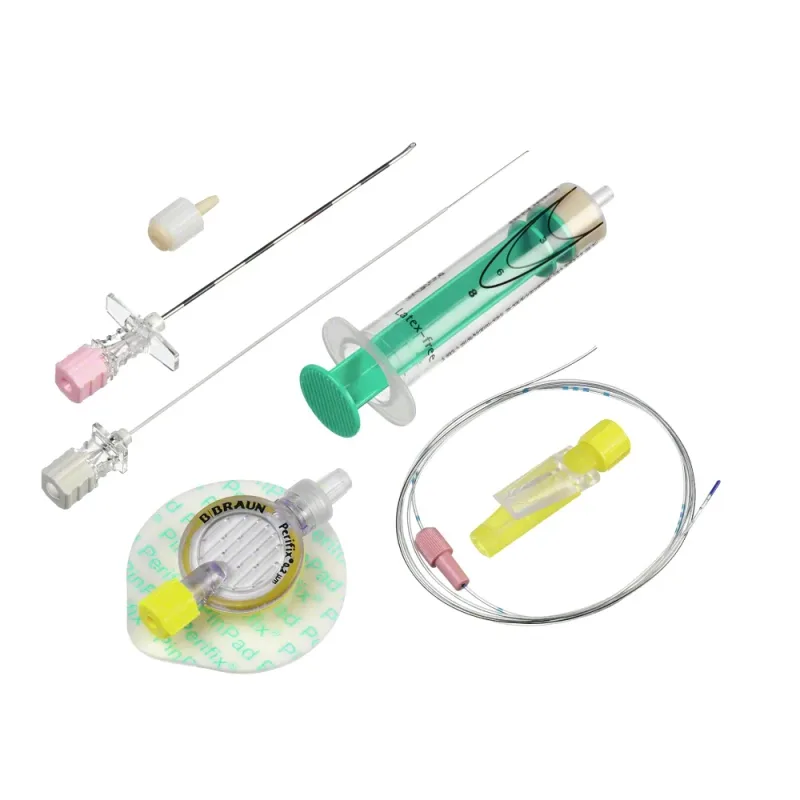
B Braun Espocan Combined Spinal Epidural Needles
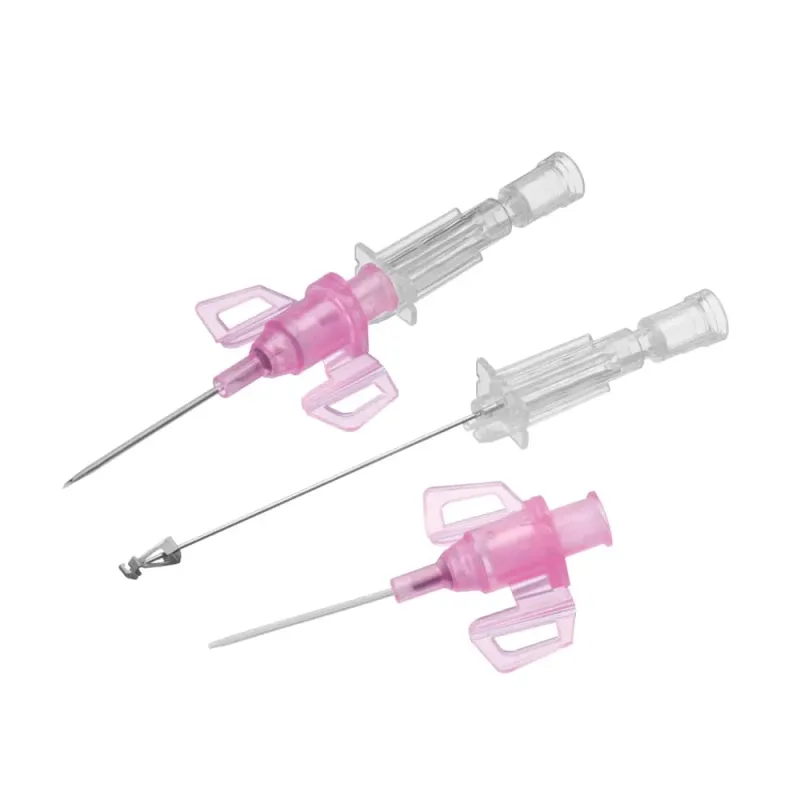
B. Braun
B Braun Introcan IV Catheter Cannula