Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
* Prices shown are indicative. Clicking "Buy" will take you to the retailer's website. Doctar may earn a small affiliate commission at no extra cost to you.
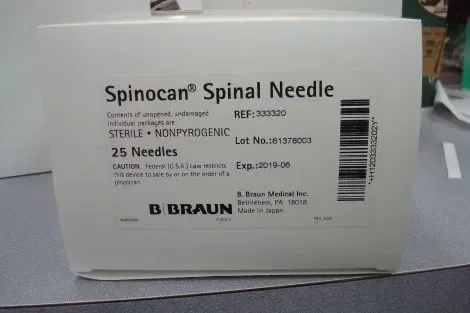
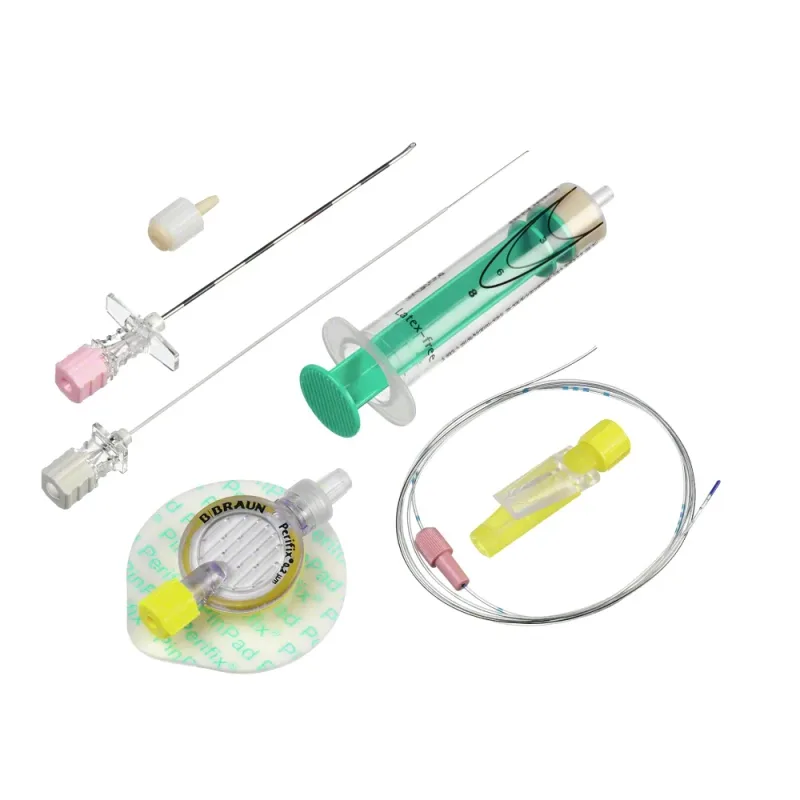
The B-Braun Spinocan Spinal Needle is a medical device designed for the administration of spinal anesthesia. It is constructed from medical-grade stainless steel and is DEHP-free, ensuring material safety. The device is intended for single-use to maintain sterility and prevent cross-contamination.
Available in packs of one or twenty-five units, it offers flexibility for various clinical settings. The needle is characterized by its Quincke-type beveled tip, engineered to provide sharpness and a low puncture force. This design aims to minimize patient discomfort and reduce tissue trauma during insertion into the subarachnoid space.
The needle is supplied sterile and should be handled and used according to aseptic techniques. Its construction and design prioritize procedural accuracy and patient safety during spinal anesthesia procedures.
The mechanism of action for the Spinocan Spinal Needle involves precise penetration of the dura mater to access the cerebrospinal fluid (CSF). The Quincke-type tip, with its specific bevel angle, is designed to create a clean puncture in the dura, facilitating the flow of CSF and reducing the likelihood of post-dural puncture headache. A key feature is the presence of a stylet within the needle lumen during insertion.
This stylet provides rigidity, preventing tissue fragments from entering the needle and obstructing the lumen. Upon reaching the target depth, the stylet is withdrawn, allowing for CSF aspiration to confirm correct placement or for the injection of anesthetic agents. The needle's design incorporates features that provide tactile feedback to the user, including a discernible sensation upon dural puncture, often referred to as a \"dura click.
\" This feedback is crucial for confirming accurate needle placement within the subarachnoid space.
Key benefits and features of the Spinocan Spinal Needle include its sharp, low-puncture force Quincke-type beveled tip, which contributes to a less traumatic insertion. The precision-ground stylet is designed to minimize tissue coring, promoting smoother passage through tissue layers. The needle's gliding characteristics are optimized to enhance the procedural experience for both the patient and the healthcare provider.
The device is available in a comprehensive range of gauges, from 18G to 27G, and lengths, typically from 80mm to 120mm, with color-coding for easy identification of different sizes. This variety ensures suitability for a wide spectrum of patient anatomies and clinical requirements. The ergonomic hub design provides enhanced control and grip for the medical professional during manipulation.
Depth markings on the needle shaft are also included to aid in accurate insertion depth estimation.
This spinal needle is utilized by a range of medical professionals, including anesthesiologists, pain management specialists, and other clinicians trained in performing spinal anesthesia and related procedures. It is also relevant for use by nurses and technicians involved in the preparation and support of these procedures. Caregivers may be involved in patient preparation and post-procedural monitoring.
Typical applications include the administration of local anesthetics for surgical procedures such as orthopedic surgery, cesarean sections, and other interventions requiring anesthesia below the waist. It is also employed in diagnostic procedures involving the spinal canal and for the management of chronic pain through intrathecal medication delivery. The device is intended for use in sterile environments, such as operating rooms, procedure suites, and pain clinics.
Important usage notes for the B-Braun Spinocan Spinal Needle include strict adherence to aseptic techniques throughout the entire procedure to prevent infection. The needle is designed for single-use only and must be discarded appropriately after use. Healthcare providers should select the appropriate needle gauge and length based on patient factors, the specific procedure, and their clinical judgment.
Confirmation of correct needle placement by observing CSF flow is a critical step before administering medication. Users should be familiar with the specific characteristics of the needle, including the tactile feedback provided upon dural puncture, to ensure safe and effective administration of spinal anesthesia. The material composition, including the use of medical-grade stainless steel and DEHP-free components, is intended to meet safety standards for medical devices.

B. Braun
B. Braun Certofix Duo Paed S 408
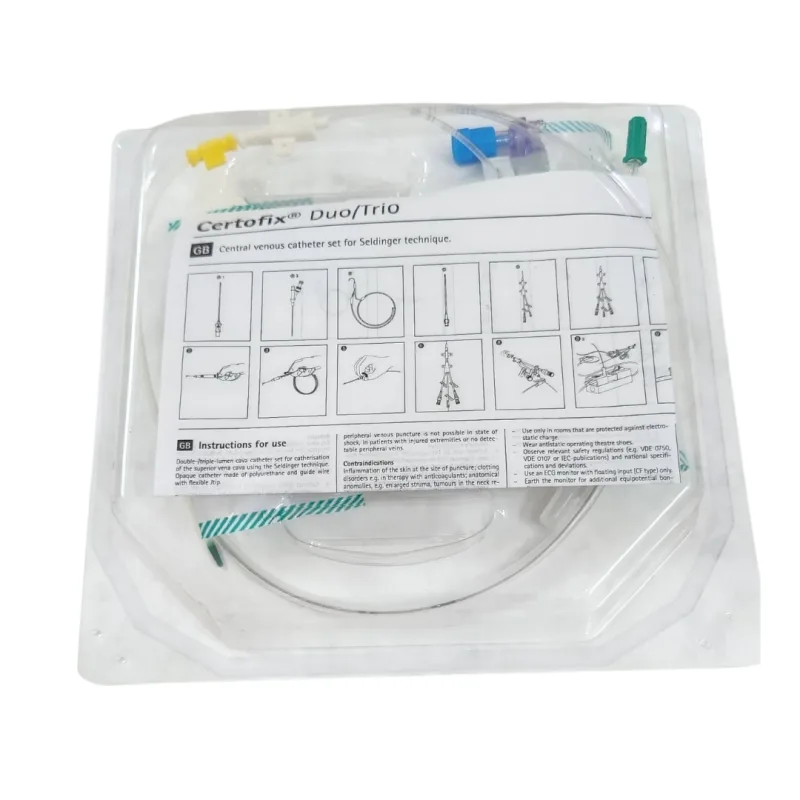
B. Braun
B. Braun Certofix Duo Central Venous Catheter Kit

B. Braun
B Braun Certofix Trio Central Venous Catheter Kit
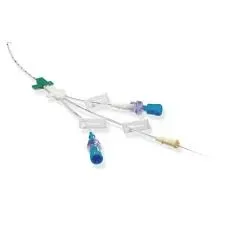
B. Braun
B. Braun Certofix Trio Paed S 508

B. Braun
B Braun Combidyn Pressure Monitoring Line, PMO Line
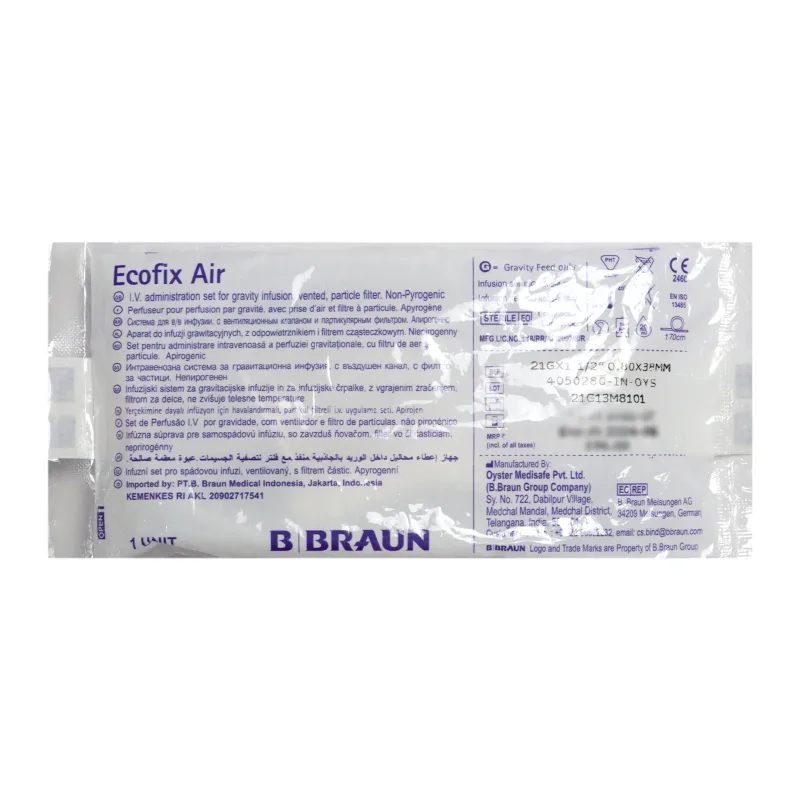
B. Braun
B Braun Ecofix Air Vented IV Infusion Set

B. Braun
B Braun Ecofix Non Vented IV Infusion Set
B. Braun
B Braun Espocan Combined Spinal Epidural Needles