Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
* Prices shown are indicative. Clicking "Buy" will take you to the retailer's website. Doctar may earn a small affiliate commission at no extra cost to you.
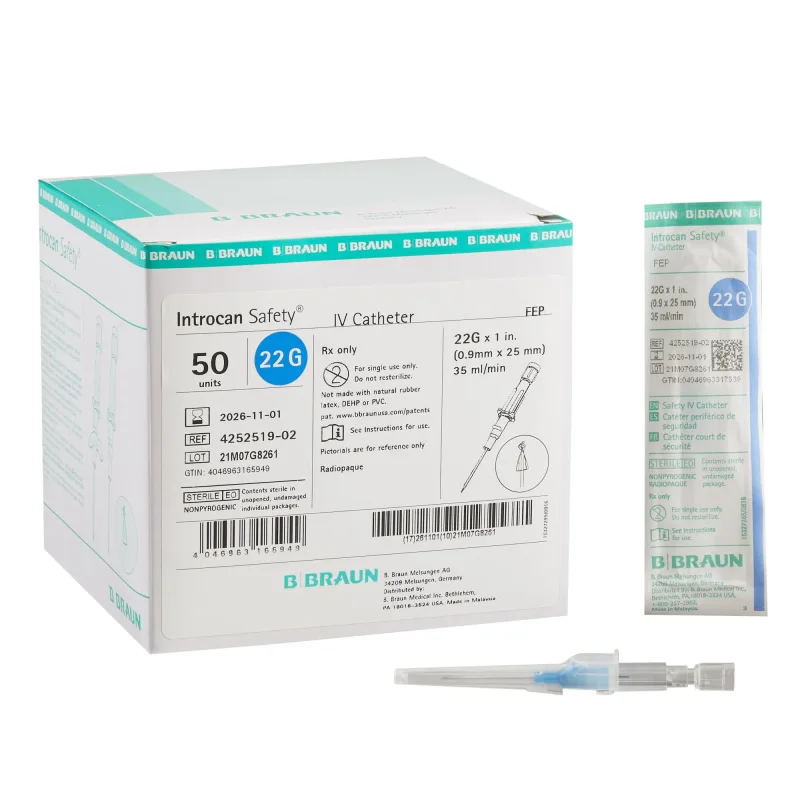
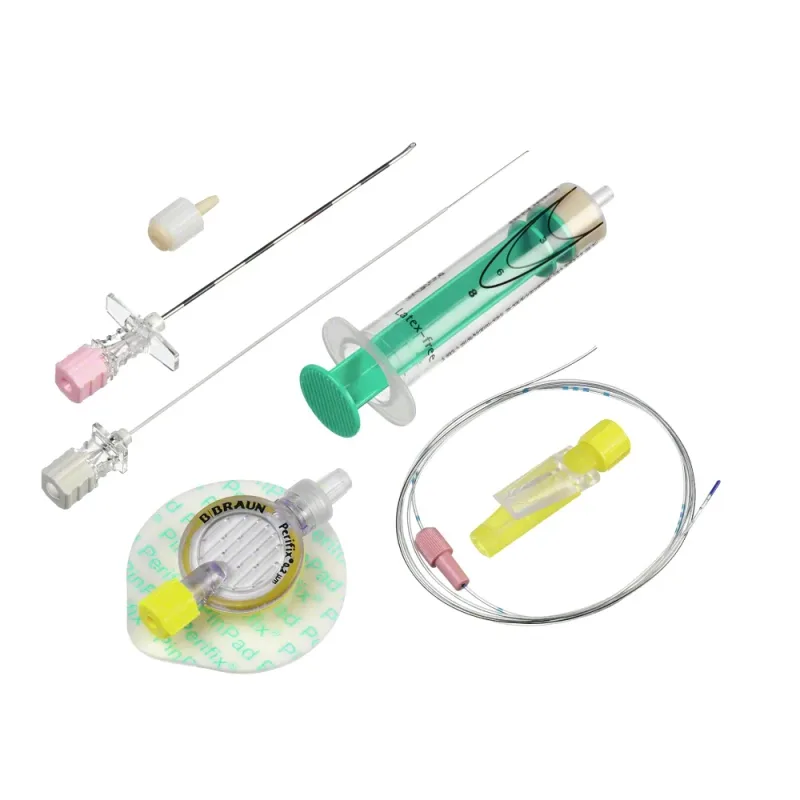
The B Braun Introcan Safety IV Cannula is a medical device designed for establishing peripheral venous access. It facilitates the safe and secure introduction of an intravenous line into a patient's vein, with an integrated safety mechanism aimed at reducing the risk of needlestick injuries for healthcare professionals. The device is constructed from materials chosen for their biocompatibility and flexibility to minimize patient discomfort and potential tissue reaction during use.
The cannula features a universal back cut bevel designed to promote smooth venipuncture and reduce the likelihood of vessel trauma. Radiopaque stripes are incorporated into the catheter material, allowing for visualization under imaging modalities if necessary. The device is also designed to be power injectable, enabling the administration of contrast media at specified flow rates, which is a critical consideration in certain diagnostic procedures.
Furthermore, the materials used in its construction are specified as not containing DEHP, Latex/Natural Rubber, or PVC, addressing potential patient sensitivities and regulatory considerations. The cannula is available in a range of gauges, typically from 14G to 24G, with corresponding color-coded hubs for easy identification of size.
The operational principle of the Introcan Safety IV Cannula centers on its passive safety mechanism. Upon activation, typically during the withdrawal of the introducer needle, the safety shield automatically covers the needle tip. This action prevents accidental needlestick exposure to healthcare providers.
The device incorporates a multi-access blood control septum, which allows for aspiration or injection through the hub without significant blood spillage, thereby minimizing patient blood loss and maintaining a cleaner working field. Double flashback technology is integrated to provide visual confirmation of successful vein cannulation. The integrated stabilization platform, often referred to as wings, aids in securing the cannula and provides a stable anchor point during infusion or aspiration.
Key features of this IV cannula include its passive safety shield, which is designed to be activated automatically, thereby reducing the cognitive load on the clinician and enhancing safety. The multi-access blood control septum is a significant benefit, offering a closed system that helps prevent blood exposure and contamination. The integrated stabilization platform provides enhanced security and ease of handling, contributing to a more reliable venipuncture.
The double flashback technology offers a dual confirmation of venous entry, increasing confidence in correct placement. The universal back cut bevel is engineered to facilitate atraumatic insertion. The presence of radiopaque stripes ensures visibility under X-ray, which can be crucial in certain clinical scenarios.
Its power injectable capability expands its utility to procedures requiring rapid or high-pressure infusions. The absence of DEHP, Latex/Natural Rubber, and PVC in its construction is a notable benefit for patients with specific allergies or sensitivities, and aligns with evolving healthcare material standards.
This IV cannula is utilized by a broad spectrum of medical professionals, including nurses, physicians, phlebotomists, and other trained healthcare providers involved in administering intravenous therapies or drawing blood. It is also relevant for caregivers in settings where intravenous access is managed. Typical applications encompass a wide range of clinical situations requiring peripheral venous access.
This includes the administration of medications, intravenous fluids, blood products, and nutritional support. It is also used for diagnostic procedures that necessitate blood sampling or the injection of contrast media for imaging studies, particularly when power injection capabilities are required. The device is suitable for use in hospital settings, outpatient clinics, emergency departments, long-term care facilities, and home healthcare environments.
Important usage notes pertain to ensuring the correct gauge and length of the cannula are selected based on the patient's vein size, the intended therapy, and the viscosity of the infusate. Proper aseptic technique must be maintained throughout the cannulation procedure to prevent infection. The safety mechanism should not be manually overridden or bypassed, as this compromises the intended safety feature.
Post-insertion care, including securement of the cannula and regular site assessment, is essential to prevent dislodgement and complications. The power injection rating should be strictly adhered to, and the cannula should not be used with flow rates exceeding its specified limits to avoid catheter damage or rupture. The device is intended for single use only and should be disposed of according to institutional protocols for medical waste.

B. Braun
B. Braun Certofix Duo Paed S 408
B. Braun
B. Braun Certofix Duo Central Venous Catheter Kit

B. Braun
B Braun Certofix Trio Central Venous Catheter Kit
B. Braun
B. Braun Certofix Trio Paed S 508

B. Braun
B Braun Combidyn Pressure Monitoring Line, PMO Line
B. Braun
B Braun Ecofix Air Vented IV Infusion Set

B. Braun
B Braun Ecofix Non Vented IV Infusion Set
B. Braun
B Braun Espocan Combined Spinal Epidural Needles