Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Learn about Severe Combined Immunodeficiency (SCID), a rare genetic disorder affecting infants' immune systems. Understand its causes, symptoms, diagnosis, and life-saving treatments like bone marrow transplants and gene therapy. Early detection is crucial for a healthy future.

Understanding Severe Combined Immunodeficiency (SCID) in Infants Severe Combined Immunodeficiency (SCID) is a rare but serious genetic disorder that affects a baby's immune system. In simple terms, it means the body's defence mechanism against infections is either absent or severely weakened. This makes infants with SCID extremely vulnerable to even common infections, which can become life-threatening very quickly. Without timely and appropriate treatment, SCID can be fatal within the first two years of a child's life. However, with early diagnosis and effective treatment, many children with SCID can lead healthy, long lives. This guide aims to provide comprehensive information about SCID for parents in India, covering its causes, symptoms, diagnosis, treatment options, and preventive measures. What is SCID? SCID is not a single disease but a group of genetic conditions. It impacts the development and function of crucial white blood cells, specifically T-cells and B-cells. These cells are the body's primary soldiers in fighting off bacteria, viruses, and other pathogens. When these cells are missing or don't work correctly, the body cannot defend itself, leaving the infant susceptible to severe and recurrent infections. The term 'bubble baby disease' was famously associated with SCID due to the story of David Vetter in the 1980s, who had to live in a sterile bubble for years to protect him from germs while doctors sought treatments. This highlights the extreme vulnerability of infants with SCID. Causes of SCID SCID is primarily an inherited condition, meaning it is passed down from parents through gene mutations. It is usually an autosomal recessive trait, meaning a child must inherit a mutated gene from both parents to develop the condition. If a person inherits only one copy of the mutated gene, they are a carrier but typically do not have SCID themselves. They can, however, pass the gene to their children. Types of SCID: X-linked SCID (XL-SCID): This is the most common form, accounting for about 50% of cases. It is caused by a mutation in a gene on the X chromosome. This type primarily affects male infants because they have only one X chromosome. Autosomal SCID: This form is caused by mutations in genes located on non-sex chromosomes. There are several subtypes of autosomal SCID, each linked to a different gene defect. It's important to note that in about 15% of SCID cases, the specific gene defect remains unknown. While some families may have a history of SCID, approximately 80% of infants born with SCID have no known family history of the disorder, meaning the mutation can occur spontaneously. SCID is rare, affecting approximately 1 in 50,000 to 60,000 newborns globally. Certain ethnic groups, such as those of Apache, Navajo, and Turkish descent, have a slightly higher incidence. Symptoms of SCID Babies with SCID may not show symptoms immediately after birth. This is because they are still protected by antibodies passed down from their mother during pregnancy. Symptoms typically begin to appear between 3 to 6 months of age, once these maternal antibodies have worn off. Common symptoms include: Frequent, persistent, and severe infections that do not respond well to treatment. Recurrent pneumonia, ear infections, or skin infections. Oral thrush (a yeast infection in the mouth) that doesn't clear up. Diarrhea that lasts for a long time. Failure to gain weight and grow properly (failure to thrive). Fever without an obvious cause. Lack of a normal immune response, such as not developing a fever when sick or not having pus formation. Swollen lymph nodes or enlarged spleen. If your baby experiences any of these symptoms, especially recurrent or severe infections, it is crucial to seek medical attention immediately. Diagnosis of SCID Early diagnosis is key to successful treatment and improving the chances of a full recovery. Fortunately, newborn screening programs are increasingly including tests for SCID. Newborn Screening: In many parts of the world, including some regions in India, newborns undergo screening shortly after birth. This screening typically measures the levels of T-cells (a type of white blood cell crucial for immunity). Low T-cell counts can indicate SCID or other immune deficiencies. Diagnostic Tests: If newborn screening suggests a potential issue, or if symptoms appear later, further tests will be conducted: Blood Tests: These tests assess the number and function of different types of immune cells (T-cells, B-cells, NK cells). Genetic Testing: This helps identify the specific gene mutation responsible for SCID, which can guide treatment decisions. Infection Screening: Tests to identify any current infections the baby may have, as these need to be treated before or alongside SCID treatment. Historically, diagnosis often occurred only after a baby had suffered their first severe infection, usually around 3 to 6 months of age. Modern screening aims to detect SCID much earlier, often within the first few days or weeks of life. Treatment for SCID The primary goal of SCID treatment is to restore a functional immune system. The most effective treatments are most successful when initiated as early as possible, ideally before the infant turns 3 months old. 1. Hematopoietic Stem Cell Transplantation (HSCT): This is considered the gold standard treatment for SCID. It involves replacing the child's faulty immune-producing stem cells with healthy ones from a donor. The ideal donor is a matched sibling. If a sibling is not available, other family members or unrelated donors may be considered. The procedure involves chemotherapy to prepare the body, followed by the infusion of healthy stem cells. With early HSCT (before 3 months), survival rates can be as high as 70% to 90%. 2. Gene Therapy: For certain types of SCID, gene therapy is a promising option. This involves correcting the genetic defect in the child's own stem cells and then reinfusing them. Gene therapy has shown significant success in treating X-linked SCID and some forms of autosomal SCID. It offers an alternative when a suitable stem cell donor is not available. 3. Enzyme Replacement Therapy (ERT): For SCID caused by a deficiency in the enzyme Adenosine Deaminase (ADA), ERT can be used. This therapy involves providing the missing enzyme to help the immune system function better. ERT is often used as a temporary measure or in conjunction with other treatments. 4. Supportive Care: While undergoing treatment, infants with SCID require meticulous supportive care to prevent and manage infections. This includes prophylactic antibiotics, antifungals, and antivirals. Isolation measures may be necessary to protect the infant from exposure to germs. Prevention and Outlook SCID itself is a genetic condition and cannot be prevented. However, its devastating effects can be mitigated through early detection and prompt treatment. Genetic Counseling: For families with a history of SCID or known carriers of the gene mutation, genetic counseling is highly recommended before planning a pregnancy. This can help assess the risk and discuss options like prenatal testing. Newborn Screening: The most critical step in preventing severe outcomes from SCID is ensuring widespread newborn screening for the condition. Early identification allows for timely intervention, significantly improving the prognosis. Outlook: The outlook for children with SCID has improved dramatically with advances in medical science. Children who receive timely treatment, especially HSCT before 3 months of age, have a good chance of developing a functional immune system and living a normal, healthy life. Without treatment, SCID is almost always fatal within the first two years. With early intervention, SCID can be considered curable for many. When to Consult a Doctor It is crucial for parents to be vigilant and consult a doctor immediately if they notice any signs of recurrent or severe infections in their infant, especially if the infections do not respond to standard treatment. This includes: Frequent fevers Persistent cough or difficulty breathing Severe diarrhea or vomiting Unexplained rashes or skin infections Oral thrush that doesn't go away Failure to gain weight or grow If your baby has undergone newborn screening and there were any concerns, follow up with your pediatrician or a specialist immediately. Frequently Asked Questions (FAQ) Q1: Is SCID contagious? No, SCID is a genetic disorder and is not contagious. It is inherited from parents through gene mutations. Q2: Can a baby with SCID be vaccinated? Live vaccines (like the MMR or varicella vaccine) are generally contraindicated for infants with SCID because their immune systems cannot handle the live virus. Inactivated vaccines may be given, but their effectiveness can be limited. Decisions about vaccinations should always be made in consultation with a pediatric immunologist. Q3: What is the long-term outlook for a child treated for SCID? With successful early treatment, most children can develop a functional immune system and lead normal, healthy lives. They may need ongoing monitoring by an immunologist, and some may have certain long-term considerations, but the quality of life can be excellent. Q4: How common is SCID in India? SCID is rare worldwide, and specific incidence rates for India are still being established. However, as screening programs expand and awareness increases, more cases are being identified. It is essential for healthcare providers and parents to be aware of the condition. Q5: Can SCID be detected before birth? Prenatal
In summary, timely diagnosis, evidence-based treatment, and prevention-focused care improve long-term health outcomes.
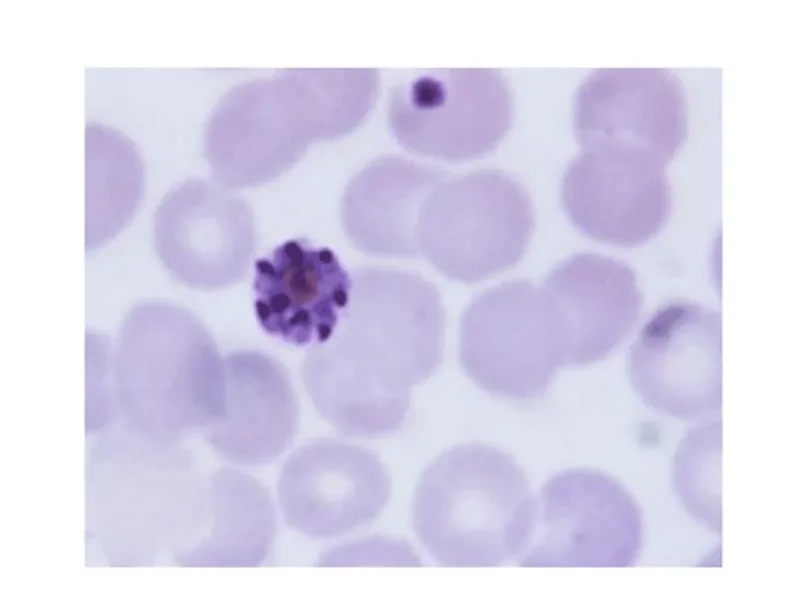
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and what results mean for patients.
April 20, 2026
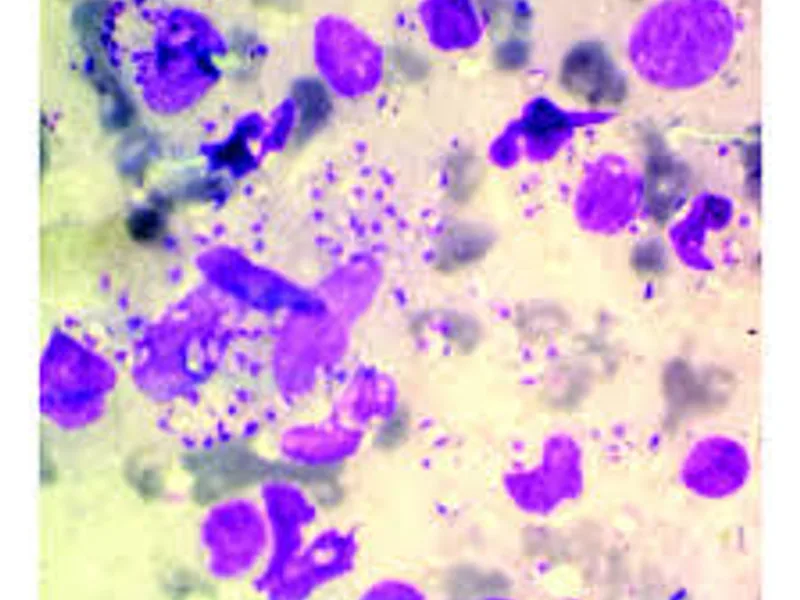
Discover how specialized staining techniques, like Giemsa, are crucial for accurately diagnosing Leishmaniasis, identifying the parasite, and guiding timely treatment. Understand this key diagnostic approach.
April 20, 2026
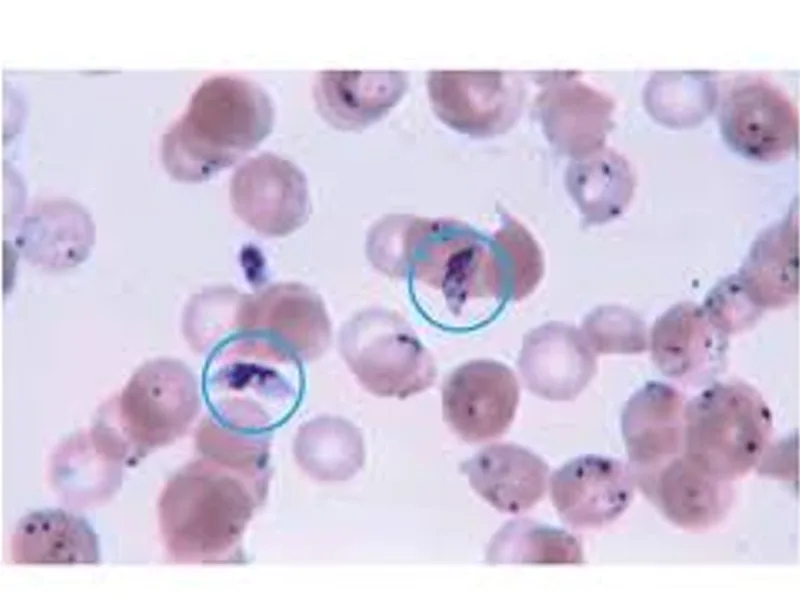
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and why it's a vital tool in combating this disease.
April 20, 2026