Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Learn about monoclonal antibody treatment for COVID-19, how it works, who is eligible, and its administration. Understand its role in fighting the virus and potential side effects.

The COVID-19 pandemic has brought about rapid advancements in medical treatments, and monoclonal antibody therapy stands out as a significant development. While many of these treatments are still under ongoing study, initial results have been highly promising in helping individuals combat the virus. This article provides a detailed look at what monoclonal antibodies are and how they have been used to treat COVID-19, offering practical insights for Indian readers.
Monoclonal antibodies are laboratory-made proteins that act like your body’s own antibodies. Antibodies are naturally produced by your immune system in response to a specific infection, such as the virus that causes COVID-19. Their primary role is to help your body fight off these infections. Monoclonal antibodies are designed to target specific parts of a virus, in this case, the spike proteins on the surface of the SARS-CoV-2 virus.
Monoclonal antibodies are administered to patients to help their bodies fight COVID-19. They work by attaching themselves to the spike proteins of the coronavirus. When these antibodies bind to the spike proteins, they effectively block the virus from entering human cells. This process slows down the infection, reduces the severity of illness, and can help prevent hospitalizations and emergency room visits. It’s crucial to understand that monoclonal antibodies are a treatment for COVID-19, not a preventive measure like a vaccine. They do not replace the need for vaccination.
Eligibility for monoclonal antibody therapy typically depends on several factors, including:
Healthcare facilities may have additional specific requirements. It is essential to consult with a healthcare professional to determine eligibility.
For pregnant or breastfeeding individuals, the decision to undergo monoclonal antibody therapy requires careful consideration. While these treatments have been used in some cases, it is highly recommended to discuss the potential risks and benefits thoroughly with an obstetrician. They can provide personalized advice based on the individual's specific health circumstances and the stage of pregnancy or breastfeeding.
Monoclonal antibody treatments are typically given intravenously (through an IV drip) at an outpatient clinic or a designated healthcare facility. The infusion process usually takes a specific amount of time, and patients are monitored by medical staff during and after the procedure. Before leaving, patients are provided with information on how to manage any potential side effects that may occur at home.
Like any medical treatment, monoclonal antibody therapy can have potential side effects. Most side effects are mild and tend to resolve on their own within a few hours. However, in rare cases, more serious reactions can occur. Common side effects may include:
Serious side effects are rare but can include severe allergic reactions. Medical staff are trained to manage these reactions. If you experience any unusual symptoms after receiving the treatment, it is important to seek medical attention promptly.
Isolation: It is crucial to continue isolating according to current local and federal guidelines even after receiving monoclonal antibody therapy. This helps prevent further spread of the virus.
Follow-up Care: Adhering to your doctor’s instructions and attending all scheduled follow-up appointments is vital for monitoring your recovery and the effectiveness of the treatment.
Consult Your Doctor: Always consult with your healthcare provider to determine if monoclonal antibody therapy is appropriate for you. They can assess your individual health status, discuss the benefits and risks, and guide you through the treatment process.
A: Yes. It is extremely important to continue isolating according to current local and federal guidelines after receiving monoclonal antibody therapy to prevent the spread of the virus.
A: No. Monoclonal antibodies are a treatment for COVID-19 and do not replace the preventive benefits of COVID-19 vaccines.
A: Monoclonal antibodies begin working as soon as they are administered and attach to the virus. Their effects in reducing viral load and preventing severe illness are typically observed relatively quickly, but recovery time can vary among individuals.
A: If you experience any side effects, especially signs of a severe allergic reaction (like difficulty breathing, swelling, or hives), seek immediate medical attention. For milder side effects, follow the advice given by the medical staff at the infusion center.
A: Monoclonal antibody treatment is designed to help your body fight the virus and reduce the severity of illness, thereby lowering the risk of hospitalization and complications. It is a supportive treatment rather than a cure, as the immune system ultimately clears the infection.
Disclaimer: This information is for educational purposes only and does not constitute medical advice. Always consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.
This section adds practical context and preventive advice to help readers make informed healthcare decisions. It is important to verify symptoms early, consult qualified doctors, and avoid self-medication for persistent health issues.
Maintaining healthy routines, following prescribed treatment plans, and attending regular checkups can improve outcomes. If symptoms worsen or red-flag signs appear, immediate medical evaluation is recommended.
Track symptoms and duration.
Follow diagnosis and treatment from a licensed practitioner.
Review medication side effects with your doctor.
Seek urgent care for severe warning signs.
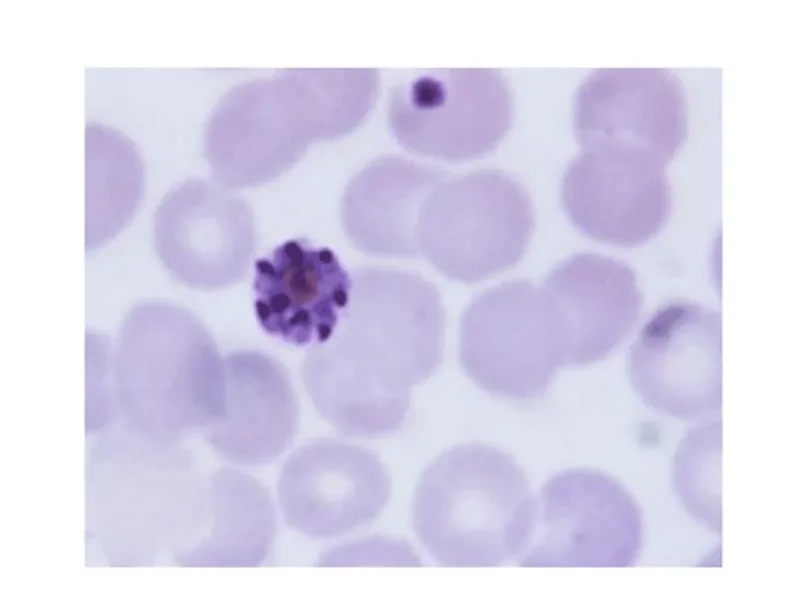
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and what results mean for patients.
April 20, 2026
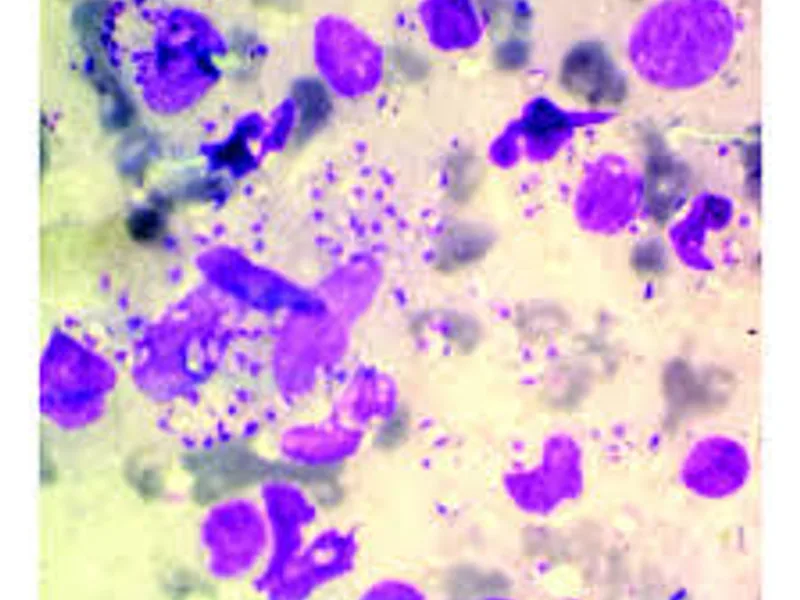
Discover how specialized staining techniques, like Giemsa, are crucial for accurately diagnosing Leishmaniasis, identifying the parasite, and guiding timely treatment. Understand this key diagnostic approach.
April 20, 2026
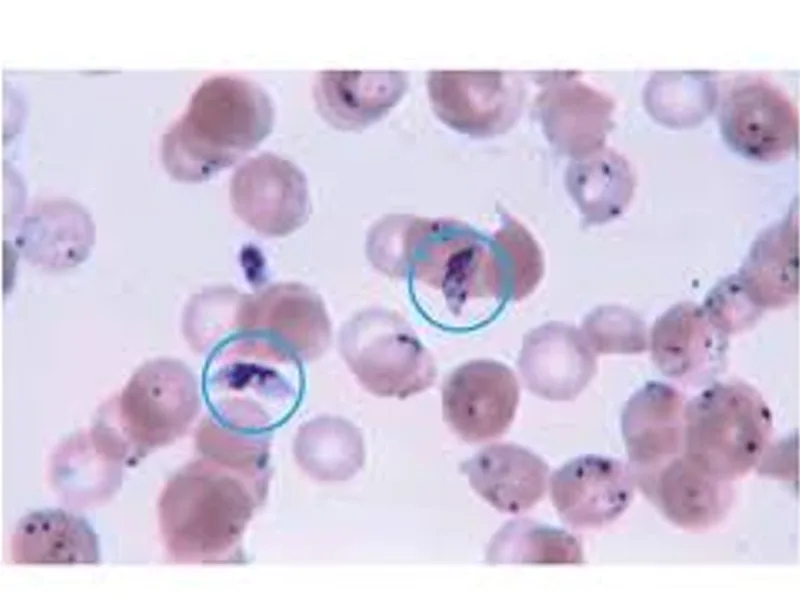
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and why it's a vital tool in combating this disease.
April 20, 2026