Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Learn about the safety and effectiveness of COVID-19 vaccines. Understand how they were developed quickly, how they work, potential side effects, and why they are a vital tool against the pandemic.

The COVID-19 pandemic has profoundly impacted our lives, and the development of vaccines has been a beacon of hope in overcoming this global health crisis. You might have questions about the safety of these vaccines, especially given their rapid development. It's natural to wonder about potential side effects, both immediate and long-term. This article aims to provide clear, reliable information about the safety and effectiveness of COVID-19 vaccines, drawing on scientific understanding and rigorous testing protocols.
The speed at which COVID-19 vaccines were developed might seem unprecedented, and it's a valid point of curiosity. Typically, vaccine development can span many years, involving sequential phases of research and testing. However, the development of COVID-19 vaccines benefited from several factors working in unison.
Scientists had been exploring and refining vaccine technologies, such as mRNA (messenger RNA), for years before the COVID-19 pandemic. This foundational research provided a significant head start. When the SARS-CoV-2 virus was identified, researchers could quickly adapt these existing platforms to target the new virus.
Governments and private organizations worldwide poured immense financial resources into vaccine research and development. This substantial funding allowed companies to conduct research, clinical trials, and manufacturing simultaneously, rather than waiting for each stage to be fully completed before starting the next. This parallel processing significantly accelerated the timeline. Furthermore, a remarkable level of global scientific collaboration meant that research findings were shared rapidly, speeding up the collective understanding of the virus and potential vaccine strategies.
While timelines were compressed, the rigorous standards for safety and effectiveness were not compromised. Clinical trials were conducted in phases, but these phases often overlapped. Importantly, companies began scaling up manufacturing while clinical trials were still underway. This was a considerable financial risk, as they could potentially lose significant investment if the vaccine proved unsafe or ineffective. However, this parallel approach meant that if the vaccine was authorized, doses were ready for distribution almost immediately.
Vaccines are a cornerstone of public health, offering protection against infectious diseases. When you receive a vaccine, your body learns to recognize and fight off a specific pathogen, like the SARS-CoV-2 virus. This prevents you from getting seriously ill if you are exposed.
Many of the widely used COVID-19 vaccines, such as those developed by Pfizer-BioNTech and Moderna, utilize mRNA technology. Here's how it works:
The mRNA itself does not enter the nucleus of your cells and does not alter your DNA. It is quickly broken down by your body after it has served its purpose.
The safety of COVID-19 vaccines has been a top priority throughout their development and deployment. Regulatory bodies like the Food and Drug Administration (FDA) in the United States and similar agencies worldwide have implemented stringent review processes.
Before any vaccine can be authorized or approved for public use, it must successfully complete three phases of clinical trials involving thousands of participants. These trials are designed to:
Even after a vaccine is authorized or approved, safety monitoring continues. Systems like the Vaccine Adverse Event Reporting System (VAERS) in the US collect reports of potential health problems following vaccination. This ongoing surveillance helps detect any rare side effects that might not have been apparent in clinical trials. Public health agencies analyze this data rigorously to ensure vaccine safety remains paramount.
Like many vaccines and medications, COVID-19 vaccines can cause side effects. Most of these are mild to moderate and short-lived, indicating that your immune system is responding and building protection.
The most frequently reported side effects include:
These symptoms typically appear within a day or two of vaccination and usually resolve within a few days. They are generally a sign that your body is building protection.
While extremely rare, some serious side effects have been identified through ongoing monitoring. These include:
It's important to remember that the risk of these rare side effects is significantly lower than the risks associated with contracting COVID-19 itself, including severe illness, hospitalization, long-term health problems, and death.
No, the COVID-19 vaccines cannot give you COVID-19. mRNA vaccines use a piece of genetic code that instructs your cells to make a harmless spike protein. They do not contain the live virus that causes COVID-19. Other vaccine types work by introducing an inactivated virus or a part of the virus, none of which can cause infection.
Yes, vaccination after infection is recommended. Natural immunity from infection can vary in strength and duration. Vaccination provides a more reliable and stronger level of protection.
Yes, COVID-19 vaccines are recommended for pregnant and breastfeeding individuals. Clinical trials and real-world data have shown them to be safe and effective during pregnancy, helping protect both the mother and the baby from severe illness.
While most side effects are mild and temporary, you should seek medical attention if you experience:
Always discuss any pre-existing health conditions or concerns with your doctor before getting vaccinated.
The development of COVID-19 vaccines represents a remarkable scientific achievement. Despite the accelerated timelines, no steps were skipped in the rigorous testing and approval processes. These vaccines are a safe and highly effective tool in our fight against the pandemic, significantly reducing the risk of severe illness, hospitalization, and death. By understanding how they work and what to expect regarding side effects, you can make an informed decision about vaccination. Staying informed with reliable sources and consulting your healthcare provider are key steps in navigating your health journey.
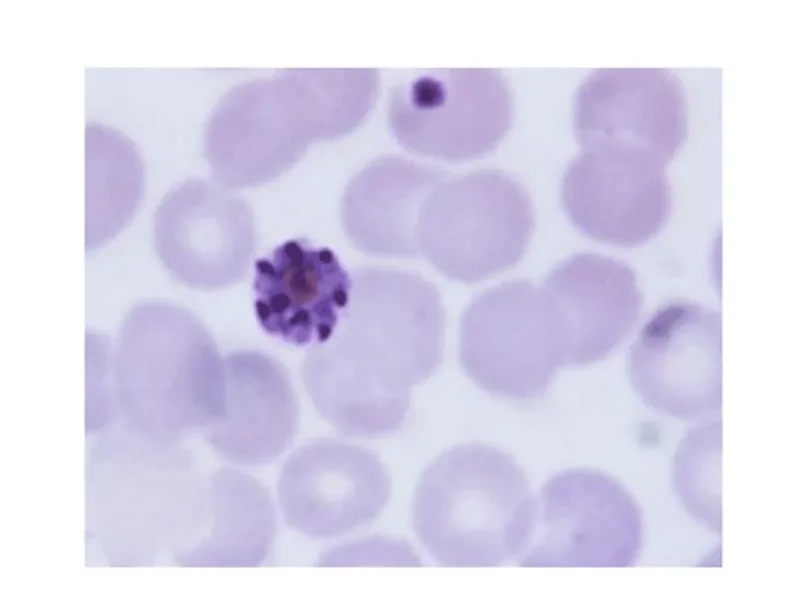
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and what results mean for patients.
April 20, 2026
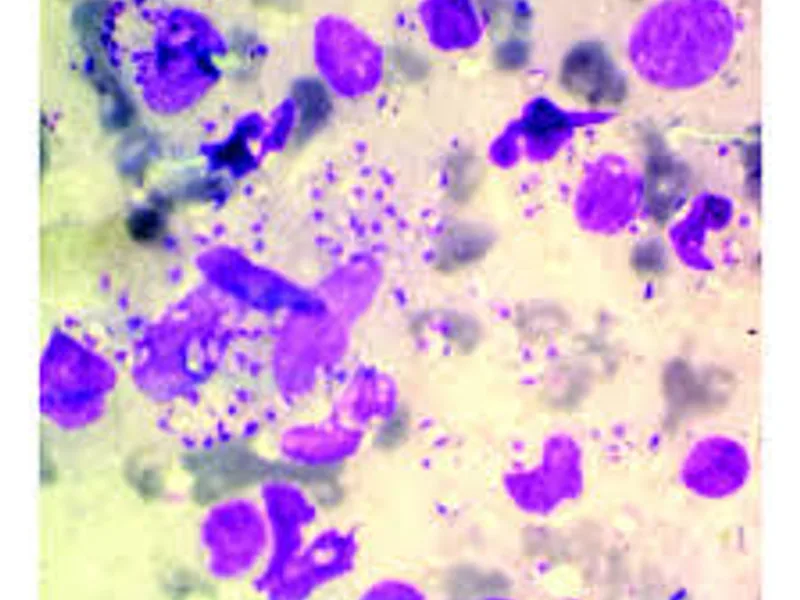
Discover how specialized staining techniques, like Giemsa, are crucial for accurately diagnosing Leishmaniasis, identifying the parasite, and guiding timely treatment. Understand this key diagnostic approach.
April 20, 2026
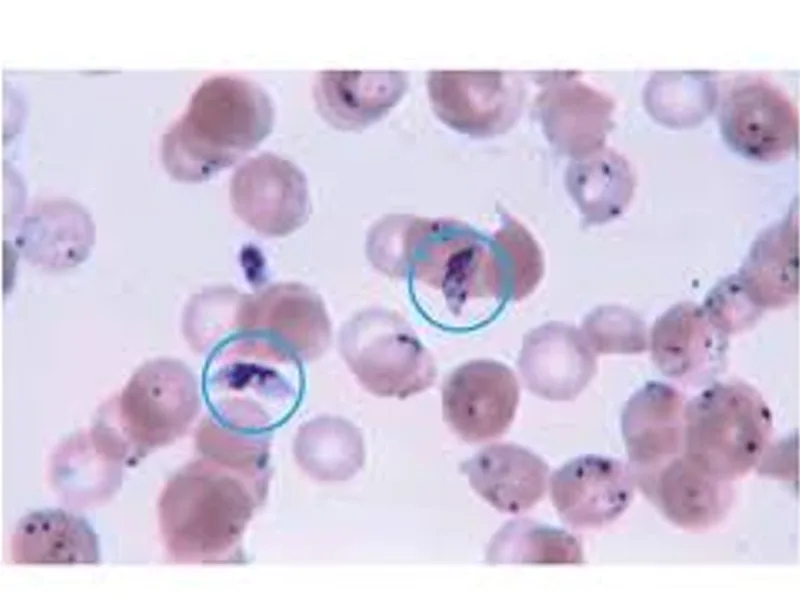
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and why it's a vital tool in combating this disease.
April 20, 2026