Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Explore how clinical trials can offer new treatment avenues for Hepatitis C. Learn about eligibility, how to find studies, and the benefits of participating in research.

If you're living with Hepatitis C, you might be wondering about the latest treatment options. While modern oral medications have made a huge difference, offering higher cure rates and fewer side effects than older treatments like interferon injections, the journey isn't over for everyone. For some, participating in clinical trials could be a pathway to accessing cutting-edge therapies and contributing to medical advancement. This guide will walk you through what clinical trials are, why they matter, and how you can find opportunities that might be a good fit for you.
Clinical trials, also known as intervention studies, are research studies that involve human volunteers. Their primary goal is to test new treatments, drugs, or medical devices to determine their safety and effectiveness. When you join a clinical trial, you're not just a passive recipient of care; you become an active partner in your own health journey. This involvement can lead to a deeper understanding of your condition and, importantly, can provide access to novel treatments that aren't yet widely available. Think of it as being at the forefront of medical discovery, helping to shape the future of Hepatitis C care.
The benefits of participating are manifold. Firstly, you gain access to potentially life-changing treatments earlier than the general public. This can be particularly significant for conditions like Hepatitis C, where advancements have been rapid. Secondly, you receive a high level of medical care and attention from a dedicated research team. Your health will be closely monitored throughout the study. Thirdly, by participating, you contribute to a greater medical understanding of diseases and their treatments. Your involvement helps researchers gather vital data that can lead to better therapies for countless others in the future.
Clinical trials are meticulously designed to yield reliable results. Researchers carefully control various factors, such as medication dosage or the effects of a new drug. One common design involves assigning participants to different groups. You might receive the experimental treatment, a placebo (an inactive substance), or a standard treatment. This comparison helps researchers understand the true impact of the intervention being studied.
Types of Studies:
The specific protocol for each trial will be clearly outlined. If you're selected for a trial, the research team will explain exactly what to expect, including medication schedules, device usage, and follow-up appointments.
Not everyone is eligible for every clinical trial. Researchers set specific criteria to ensure the safety of participants and the validity of the study's results. These criteria fall into two main categories:
These are the requirements you must meet to be considered for a trial. They often relate directly to the condition being studied. For Hepatitis C trials, this might include:
These are factors that would prevent you from participating, even if you meet the eligibility criteria. Exclusion criteria are primarily in place for safety reasons or to avoid confounding results. Common exclusion criteria include:
It's vital to have an open conversation with your doctor about these criteria. They can help you understand if you meet the requirements and if participation is a safe and practical option for you.
So, how do you actually find these trials? Several resources can help you navigate the landscape of clinical research:
Your hepatologist or gastroenterologist is your first and most important point of contact. They are often aware of ongoing studies in their network or can refer you to specialists who are involved in research. Discuss your interest and inquire if they know of any trials that might be suitable for your specific situation.
Several reputable online platforms list clinical trials. These databases allow you to search by condition, location, and other criteria:
Organizations dedicated to Hepatitis C awareness and support often have information about clinical trials or can direct you to resources. They can be a valuable source of both information and emotional support.
Participating in a clinical trial is a commitment. Beyond the medical aspects, consider these practical factors:
The progress in Hepatitis C treatment has been nothing short of remarkable. From lengthy interferon injections with moderate success rates to highly effective, short-course oral medications, the landscape has transformed. Clinical trials continue to play a pivotal role in this evolution. They are exploring new drug combinations, investigating ways to simplify treatment further, and studying the long-term outcomes of cure. By participating, you are not only potentially benefiting yourself but also becoming a vital part of the ongoing effort to eradicate Hepatitis C and improve the lives of millions.
If you have Hepatitis C and are interested in exploring treatment options, including clinical trials, it's essential to consult your doctor. They can provide personalized advice based on your health status, discuss the risks and benefits of different approaches, and help you identify suitable research opportunities. Don't delay seeking medical advice; early intervention and informed decisions are key to managing Hepatitis C effectively.
In many trials, you will receive the experimental drug. However, some trials compare the new drug to a placebo or a standard treatment. The study protocol will clearly state which group you might be assigned to.
Safety is the top priority. All trials are rigorously reviewed by ethics committees and regulatory bodies. Participants are closely monitored, and potential risks are explained thoroughly. You have the right to withdraw at any time.
Depending on the trial, you might continue to receive the study drug for a period, transition to standard care, or be referred back to your regular doctor. The post-trial plan will be discussed with you before you enroll.
Some trials are for newly diagnosed patients, while others may include individuals who have not responded to previous treatments or who are experiencing long-term effects. Your eligibility will depend on the specific trial criteria.
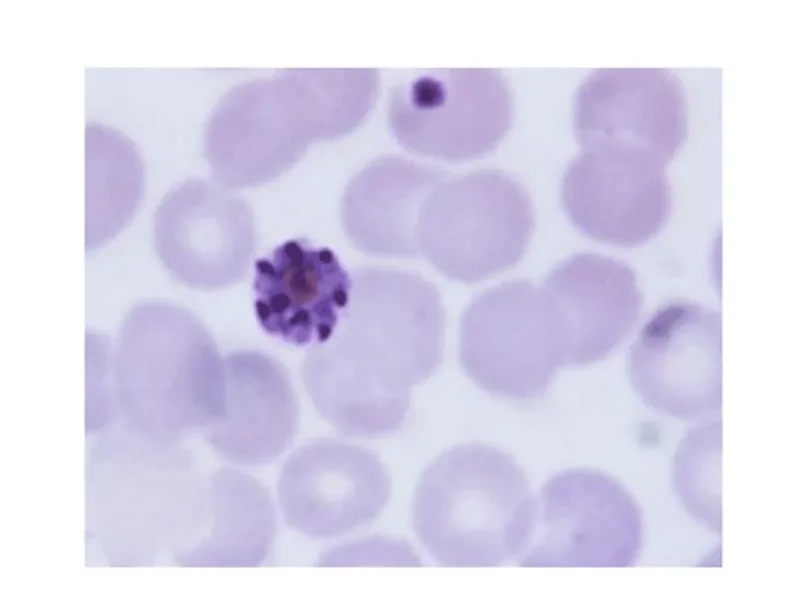
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and what results mean for patients.
April 20, 2026
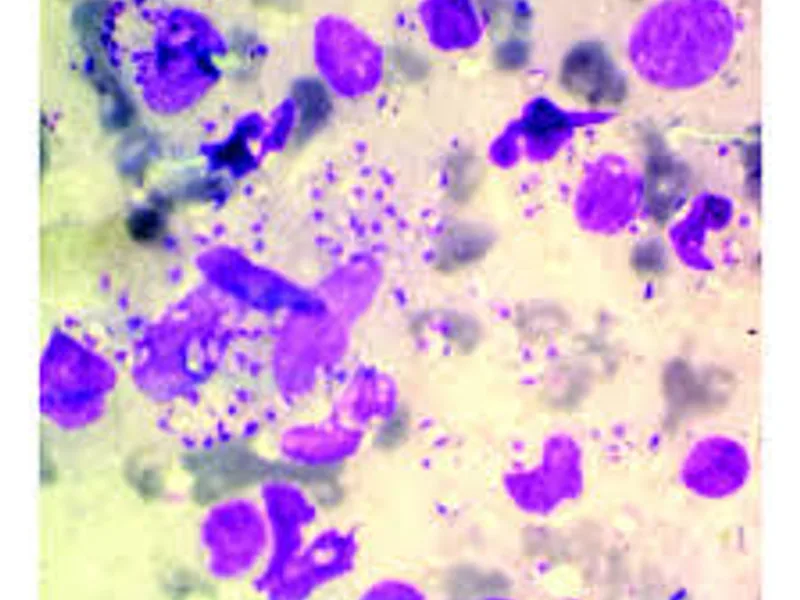
Discover how specialized staining techniques, like Giemsa, are crucial for accurately diagnosing Leishmaniasis, identifying the parasite, and guiding timely treatment. Understand this key diagnostic approach.
April 20, 2026
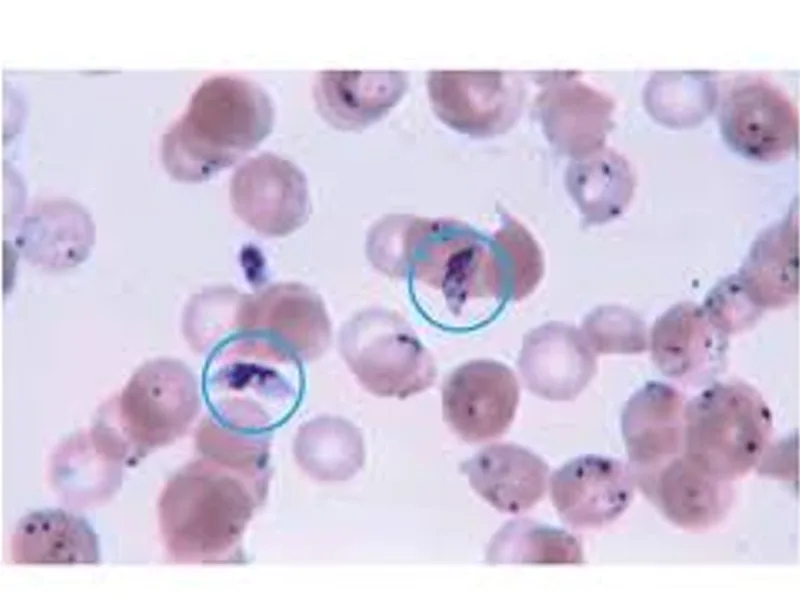
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and why it's a vital tool in combating this disease.
April 20, 2026