Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
A comprehensive review of the BinaxNOW COVID-19 Antigen Self-Test, covering its features, accuracy, usage, and considerations for Indian users. Learn how to use this at-home testing kit effectively.

The COVID-19 pandemic has reshaped our lives, and with it, the importance of accessible and reliable testing has become paramount. For individuals in India seeking to monitor their health or comply with travel and workplace requirements, understanding at-home testing options is crucial. The BinaxNOW COVID-19 Antigen Self-Test, an FDA-authorized kit, offers a convenient way to detect active SARS-CoV-2 infections. This review delves into its features, accuracy, usage, and considerations specifically for the Indian context.
The BinaxNOW COVID-19 Antigen Self-Test is an over-the-counter diagnostic tool designed to detect the presence of specific proteins (antigens) from the SARS-CoV-2 virus, the virus responsible for COVID-19. It is authorized by the U.S. Food and Drug Administration (FDA) for emergency use, meaning it has undergone rigorous evaluation for its intended purpose. This test is suitable for individuals experiencing symptoms of COVID-19, as well as those who may have been exposed to the virus but are asymptomatic.
As recommended by health authorities globally, including the Centers for Disease Control and Prevention (CDC), frequent testing plays a vital role in curbing the spread of COVID-19. This is particularly true in densely populated areas like India, where the virus can spread rapidly. Regular testing helps in early detection, enabling prompt isolation and treatment, thereby protecting vulnerable populations and preventing widespread outbreaks. If you have been in close contact with someone who has tested positive or are experiencing symptoms, testing becomes even more critical.
The BinaxNOW test utilizes a shallow nasal swab to collect a sample. This sample is then mixed with a solution and applied to a test card. The test card contains antibodies that will react with viral antigens if they are present in the sample. The results are typically displayed as lines on the card within a specific timeframe. It’s important to note that this is a rapid antigen test, which detects active viral infections.
A typical BinaxNOW COVID-19 Antigen Self-Test kit, designed for two tests, contains:
While the kit is comprehensive, you will need a reliable timer or your smartphone to track the incubation period for accurate results.
Using the BinaxNOW test is designed to be straightforward. Always refer to the specific instructions included in your kit, but here’s a general overview:
The interpretation of the results is based on the presence or absence of lines:
Important Note on Testing Frequency:
Studies suggest that the BinaxNOW test can be reliable in detecting variants of concern, including Omicron and Delta. However, like all antigen tests, its sensitivity can be influenced by the stage of infection and viral load. For individuals with very early or very low viral loads, a negative result does not entirely rule out infection. In such cases, or if symptoms develop despite a negative result, confirmatory testing (like RT-PCR) or consultation with a doctor is recommended.
The BinaxNOW test is designed for self-administration and is generally safe when used as directed. Ensure you follow the instructions carefully to avoid contamination or incorrect sample collection. Dispose of used test components properly according to local guidelines.
While the BinaxNOW test is FDA-authorized, its availability and pricing in India may vary. It's advisable to check with local pharmacies, online medical supply stores, or authorized distributors for current availability and cost. Prices can fluctuate based on import duties, supplier markups, and market demand.
Some users have reported issues such as receiving only one test kit when the box was advertised to contain two, or receiving products with compromised seals. It is essential to purchase from reputable sources and inspect the packaging upon arrival. If you encounter any discrepancies, contact the seller or manufacturer immediately.
In countries where it is widely available and regulated, insurance coverage for COVID-19 tests can vary. For users in India, it is crucial to check with your health insurance provider regarding coverage for over-the-counter diagnostic kits like BinaxNOW. Policies and government regulations regarding COVID-19 testing coverage may differ.
It is essential to consult a healthcare professional in the following situations:
Yes, the BinaxNOW test can be used by individuals without symptoms. However, if you are asymptomatic, the manufacturer recommends testing twice over three days with at least 24 hours between tests for a more reliable assessment.
Antigen tests like BinaxNOW are generally less sensitive than RT-PCR tests, especially at lower viral loads. While BinaxNOW is effective at detecting active infections, a negative result, particularly in asymptomatic individuals, might not completely rule out infection. RT-PCR tests are considered the gold standard for COVID-19 diagnosis due to their higher sensitivity.
No, each swab and test card is designed for single use only. Reusing them will lead to inaccurate results.
If your test result is invalid, it means the test did not work correctly. You should discard the used kit and repeat the test using a new BinaxNOW kit, ensuring you follow all instructions precisely.
Store the kit at room temperature (between 2°C and 30°C or 36°F and 86°F) in its original, sealed pouch until ready to use. Avoid freezing or exposing it to excessive heat.
The BinaxNOW COVID-19 Antigen Self-Test offers a convenient and accessible option for at-home COVID-19 testing in India. By understanding its usage, limitations, and interpreting results correctly, individuals can make informed decisions about their health and contribute to controlling the spread of the virus. Always remember to consult healthcare professionals for diagnosis, treatment, and personalized medical advice.
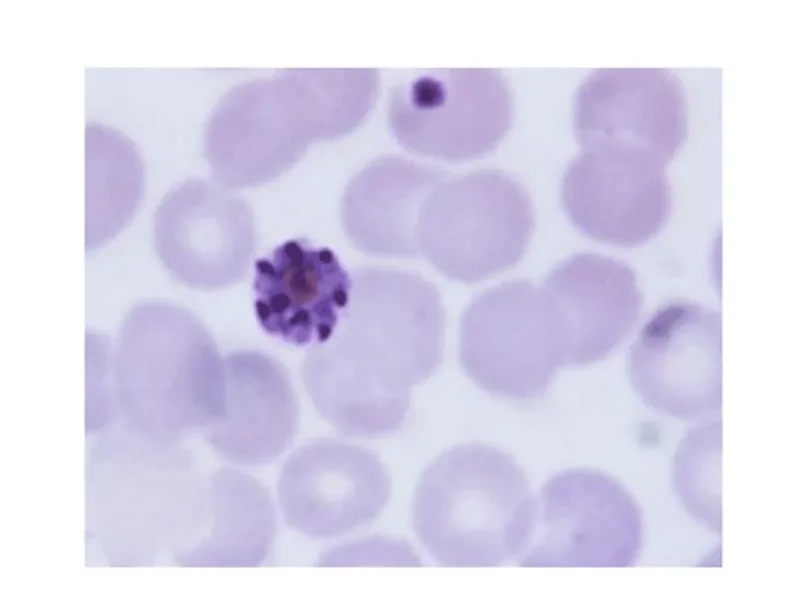
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and what results mean for patients.
April 20, 2026
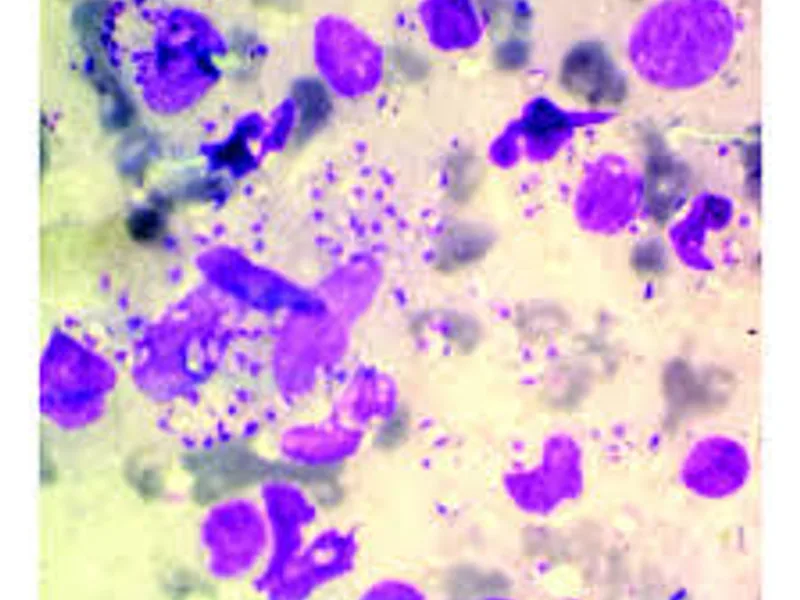
Discover how specialized staining techniques, like Giemsa, are crucial for accurately diagnosing Leishmaniasis, identifying the parasite, and guiding timely treatment. Understand this key diagnostic approach.
April 20, 2026
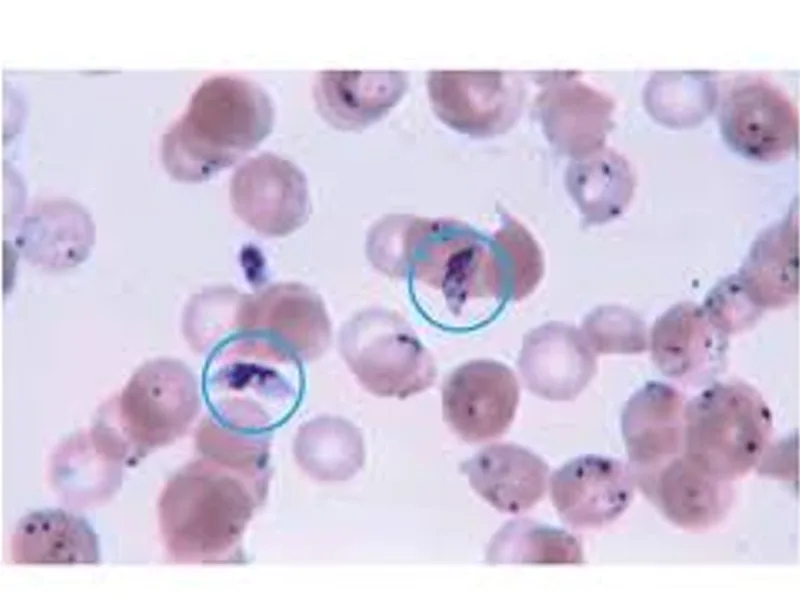
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and why it's a vital tool in combating this disease.
April 20, 2026