Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Discover Apretude, the revolutionary injectable medication for HIV prevention. Learn how this every-two-month injection offers a convenient and highly effective alternative to daily pills, potentially transforming HIV prevention strategies in India.

HIV is a global health challenge, and in India, awareness and proactive prevention are key. For years, daily oral pills like Truvada have been the cornerstone of HIV prevention, a strategy known as Pre-Exposure Prophylaxis (PrEP). While effective, daily adherence can be a significant hurdle for many. But what if there was a way to prevent HIV that didn't involve remembering a pill every single day? Enter Apretude, a groundbreaking injectable medication that's set to change the game in HIV prevention.
The U.S. Food and Drug Administration (FDA) approved Apretude in December 2021, marking a monumental step forward. This isn't just another pill; it's an injectable medication designed to be administered every two months. Imagine the freedom! No more daily reminders, no more worries about missed doses. This could be a game-changer, especially for individuals who find daily medication regimens challenging.
Apretude is a long-acting injectable formulation of cabotegravir. It belongs to a class of drugs called integrase strand transfer inhibitors (INSTIs). In simple terms, it works by preventing the HIV virus from multiplying in your body. When taken as prescribed, it significantly reduces the risk of acquiring HIV through sexual contact.
The effectiveness of Apretude has been demonstrated in two major clinical trials. These trials compared Apretude to Truvada, the daily oral PrEP medication. The results were compelling:
These figures highlight the immense potential of Apretude as a powerful tool in the fight against HIV. It's not just about reducing risk; it's about offering a highly effective alternative that can fit seamlessly into people's lives.
Let's talk about the elephant in the room: adherence. For any medication to be truly effective, it needs to be taken consistently. Daily pills, as effective as they are, can be forgotten. Life gets busy, schedules change, and sometimes, taking that pill at the right time slips through the cracks. This is where Apretude shines.
The ability to receive an injection every two months, administered by a healthcare professional, addresses this adherence challenge head-on. Dr. David Rosenthal, an immunologist who has been a strong advocate for PrEP, points out the significant advantage: “The advantage of [Apretude] is that it is a medication that can be given just once every 2 months in the doctor’s office, and it is a way to make sure that you don’t have to take the medication on a daily basis.” This convenience could dramatically improve the number of people who are consistently protected against HIV.
Apretude is indicated for HIV-negative adults and adolescents at risk of sexually acquired HIV infection. It's particularly beneficial for:
It's important to remember that Apretude is a *prevention* medication. It is not a cure for HIV and should only be prescribed to individuals who are confirmed to be HIV-negative immediately before starting the medication and before each injection.
Like any medication, Apretude can have side effects. The clinical trials reported a higher incidence of side effects with Apretude compared to Truvada. These commonly include:
While these side effects are generally manageable, it's essential to discuss any concerns with your healthcare provider. The FDA has also included a boxed warning, emphasizing the need for confirmed negative HIV tests before initiating and during treatment to prevent the development of drug-resistant HIV.
Before you can even consider Apretude, a thorough medical evaluation is necessary. Your doctor will perform tests to confirm that you are indeed HIV-negative. Regular HIV testing will also be required throughout your treatment with Apretude.
You should consult a doctor immediately if:
Early diagnosis and consistent medical care are paramount in managing and preventing HIV.
The approval of Apretude represents a significant leap forward in HIV prevention. While it's currently approved in the U.S., its potential impact on global HIV prevention efforts, including in India, is immense. By offering a more convenient and potentially more effective option, Apretude could help reduce new HIV infections and bring us closer to ending the epidemic.
The journey to a world without HIV requires a multi-pronged approach, and innovative medications like Apretude are vital components of that strategy. We need to ensure that such advancements are accessible and understood by communities across India. Open conversations, robust awareness campaigns, and accessible healthcare services will be crucial in harnessing the full potential of this revolutionary injectable PrEP.
The main advantage is the convenience of an injection administered every two months, which can significantly improve adherence compared to taking a daily pill.
No, Apretude is a medication for HIV *prevention* (PrEP). It is not a cure for HIV.
No, Apretude should only be prescribed to individuals who are confirmed to be HIV-negative. Taking it if you have HIV can lead to drug-resistant HIV.
Common side effects include reactions at the injection site, headache, fatigue, fever, muscle pain, back pain, and rash.
Apretude is administered as an injection every two months after an initial, more frequent dosing schedule.
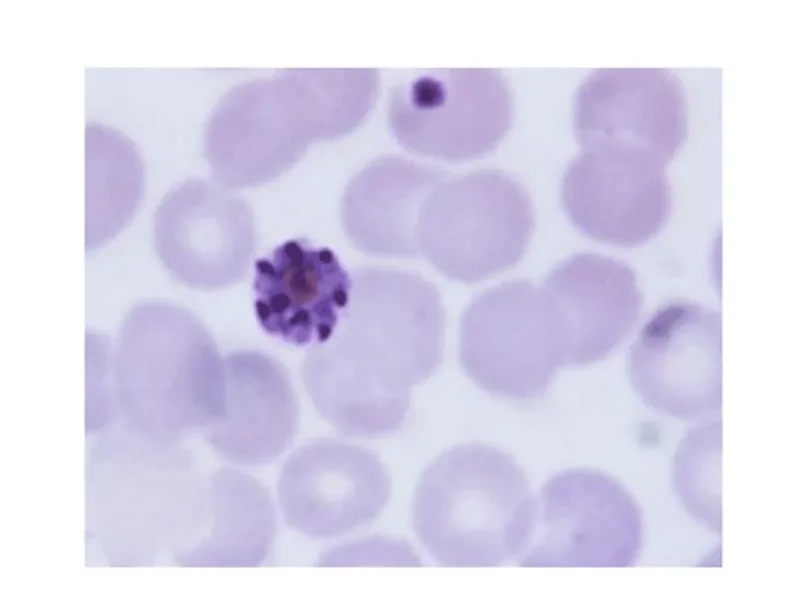
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and what results mean for patients.
April 20, 2026
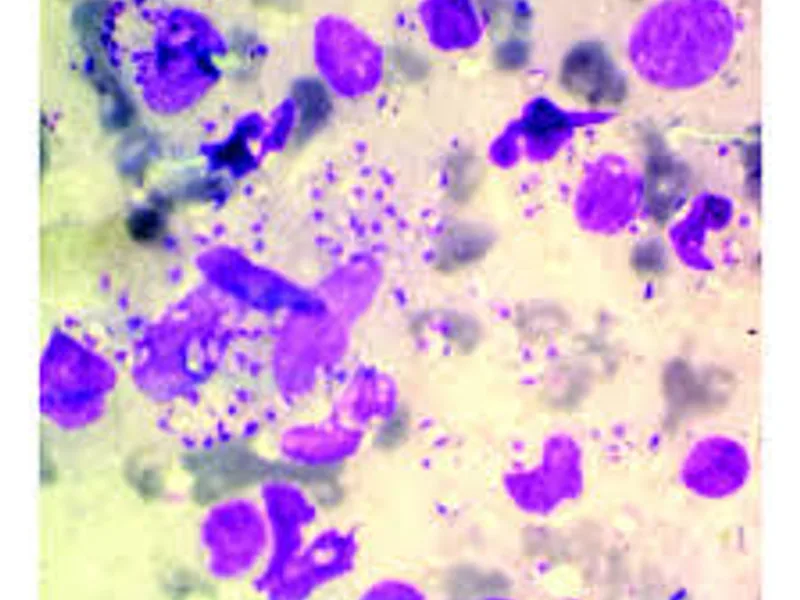
Discover how specialized staining techniques, like Giemsa, are crucial for accurately diagnosing Leishmaniasis, identifying the parasite, and guiding timely treatment. Understand this key diagnostic approach.
April 20, 2026
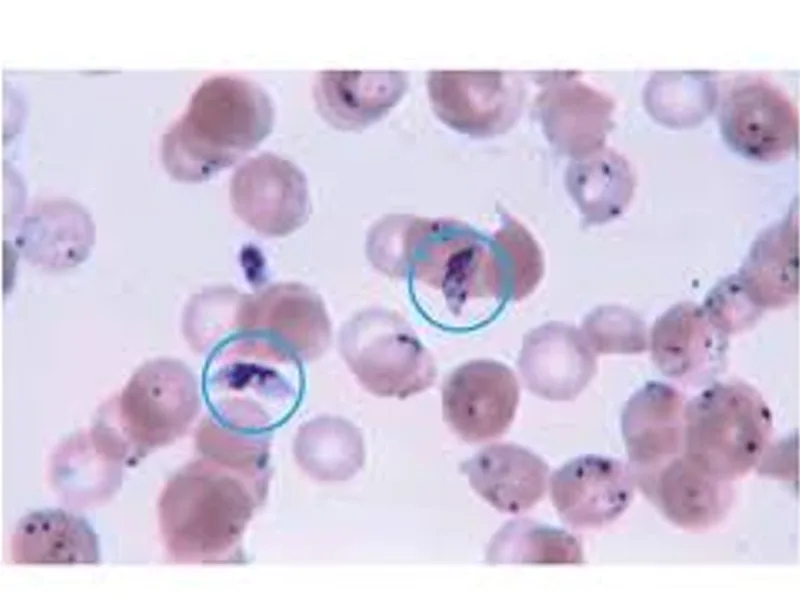
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and why it's a vital tool in combating this disease.
April 20, 2026