Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Rubbing alcohol and hydrogen peroxide are common household disinfectants. Learn their differences, how they work, and when to use each for effective germ-killing at home.

In many Indian households, keeping surfaces clean and germ-free is a top priority. You've probably reached for a bottle of rubbing alcohol or hydrogen peroxide countless times to disinfect cuts, clean countertops, or wipe down high-touch areas. Both are common, readily available disinfectants, but do you know which one to use when, and for what purpose? Understanding their differences can help you make smarter choices for a healthier home and family. This guide will break down how these two popular disinfectants work, their strengths, their weaknesses, and how to use them effectively.
Let's start with the basics. You might be surprised to learn they are quite different chemically, even though they both aim to tackle germs.
Rubbing alcohol, commonly found in medicine cabinets, typically contains isopropanol, also known as isopropyl alcohol. The bottles you buy usually have a concentration between 60% and 80% isopropanol mixed with water. It's a go-to for many because it's effective against a wide range of common germs, including bacteria, viruses, and fungi. In fact, at concentrations of 70% or higher, isopropanol is known to effectively kill the virus that causes COVID-19. Its quick evaporation makes it convenient for disinfecting skin and surfaces where rapid drying is desired.
Hydrogen peroxide, with the chemical formula H2O2, is not an alcohol at all. Its structure is very similar to water (H2O), but it has an extra oxygen atom. This additional oxygen atom makes it a potent oxidizer, meaning it can break down organic matter and kill microorganisms. The typical pharmacy bottle contains a 3% solution of hydrogen peroxide in water. This concentration is strong enough to tackle bacteria, viruses, fungi, and even tough spores. Like rubbing alcohol, it can also destroy the SARS-CoV-2 virus. Beyond disinfection, hydrogen peroxide has a multitude of other uses around the house, from stain removal to cleaning jewelry.
This is the million-dollar question, and the answer isn't a simple 'one is always better.' Their effectiveness depends heavily on a few factors:
Let's break down their performance in different scenarios.
When it comes to cleaning minor cuts, scrapes, or sterilizing skin before injections, rubbing alcohol is generally the preferred choice. Why? Because hydrogen peroxide, while a powerful disinfectant, can be too harsh for direct application on skin. It can sometimes irritate the skin, delay healing, or even damage healthy tissue if used improperly. Rubbing alcohol, when used appropriately, evaporates quickly and is less likely to cause significant irritation. Always use a concentration of 70% or higher and avoid diluting it.
Real-life scenario: Little Priya has a scraped knee after falling off her bicycle. Her mother quickly reaches for the rubbing alcohol to gently clean the wound before applying a bandage. She knows it will help kill any lingering germs without causing too much discomfort for Priya.
Both rubbing alcohol and hydrogen peroxide can be effective on hard, non-porous surfaces like countertops, doorknobs, stainless steel, and glass. However, there are important considerations for each:
When it comes to fabrics, things get a bit trickier:
Here’s a simple way to remember:
No matter which disinfectant you choose, always prioritize safety:
Yes, a 70% isopropyl alcohol solution can be used to disinfect phone screens. However, avoid getting excessive liquid into ports or openings. Wipe gently and allow it to air dry. Check your phone manufacturer's recommendations, as some advise against alcohol-based cleaners.
While 3% hydrogen peroxide is a disinfectant, it's not typically the first choice for cleaning baby bottles due to its taste and potential for residue if not rinsed thoroughly. Hot, soapy water and thorough rinsing are usually recommended. For sterilization, boiling or using a steam sterilizer is preferred.
Rubbing alcohol is a broad-spectrum disinfectant and kills most common bacteria, viruses, and fungi. However, it's not effective against all types of germs, such as bacterial spores (like C. difficile).
No, for effective disinfection, use the 3% solution as is. Diluting it will significantly reduce its germ-killing power.
No, you don't need to wipe off hydrogen peroxide. It breaks down into water and oxygen, leaving no harmful residue. Simply let it sit for at least 10 minutes.
Choosing the right disinfectant for the job ensures maximum effectiveness and safety. By understanding the properties of rubbing alcohol and hydrogen peroxide, you can confidently maintain a clean and healthy environment for your family.
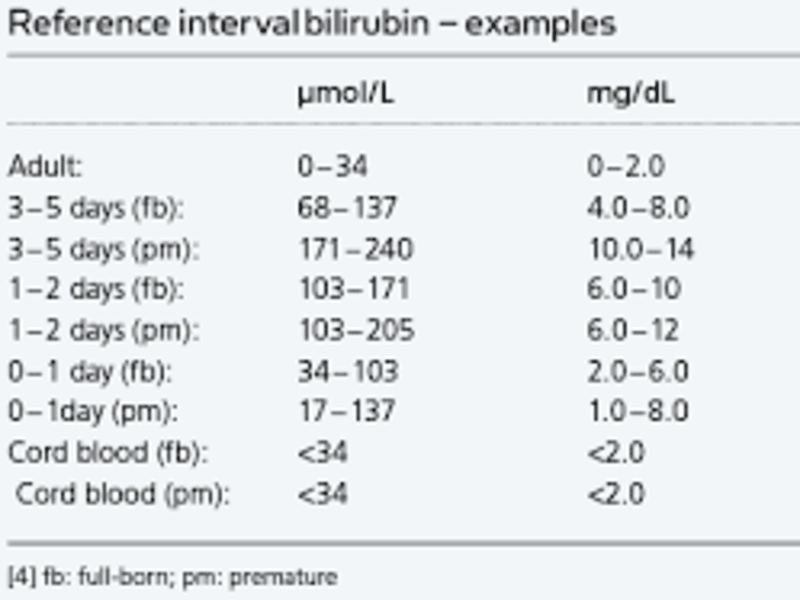
Bilirubin levels help assess liver health. This guide explains normal bilirubin values, types, causes of abnormal levels, symptoms, and when to seek medical advice.
April 11, 2026

Learn how to properly clean and care for your new ear piercing to ensure it heals beautifully and avoid infection. Follow our step-by-step guide for optimal results.
April 1, 2026

Discover practical, science-backed tips to sharpen your reaction time for gaming, sports, and everyday life. Learn how to improve your brain's speed and accuracy.
April 1, 2026