Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
* Prices shown are indicative. Clicking "Buy" will take you to the retailer's website. Doctar may earn a small affiliate commission at no extra cost to you.
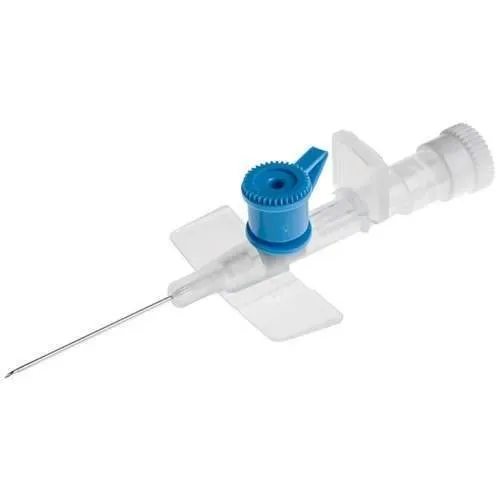
The BD Venflon I IV Cannula is a peripheral intravenous catheter designed for establishing and maintaining venous access. It facilitates the administration of fluids, medications, and blood products, as well as the withdrawal of blood samples. The device is constructed from medical-grade stainless steel for the needle component and a biocompatible polymer for the catheter.
Available in gauges of 18, 20, and 22, this cannula is intended for use by trained medical professionals in various healthcare settings. The product is offered in individual packs or in bulk quantities of 50 units.
This IV cannula incorporates a passive safety mechanism to mitigate the risk of needlestick injuries and blood exposure during use. Upon withdrawal of the needle, an integrated safety feature automatically shields the needle tip, preventing accidental sharps injuries. The device also features BD Instaflash Needle Technology, which provides immediate visual confirmation of successful vessel entry.
This technology aims to enhance the likelihood of achieving venous access on the initial attempt, thereby potentially reducing patient discomfort and the number of venipuncture procedures required.
The catheter material, identified as BD Vialon, is engineered to soften upon insertion into the vein, conforming to the vessel's contours. This characteristic is intended to reduce mechanical phlebitis and may allow for extended dwell times, potentially leading to fewer catheter changes and improved patient outcomes. The cannula's design includes a back-cut needle for optimized penetration of both the skin and the vein wall, promoting a smoother insertion process.
The winged hub provides a secure grip for healthcare providers, aiding in precise manipulation during insertion and stabilization.
This device is utilized by a range of medical professionals, including nurses, physicians, and phlebotomists, across diverse clinical environments. Typical applications include general ward care, emergency departments, intensive care units, and outpatient clinics where intravenous therapy is required. It is suitable for patients of all ages requiring short to medium-term venous access.
The snap cap port design is intended to prevent inadvertent opening, contributing to a secure and hygienic intravenous connection. A removable white vent cap offers additional flexibility for specific clinical protocols.
Important usage notes include adherence to aseptic techniques during insertion and maintenance of the IV line to prevent infection. The selection of the appropriate cannula gauge should be based on the patient's vein size, the intended therapy, and the required flow rate. Healthcare providers should ensure the passive safety mechanism is activated correctly after needle withdrawal.
The softening properties of the catheter material should be considered when planning for long-term venous access. Proper securement of the catheter wings is essential to prevent dislodgement and maintain the integrity of the IV site.

BD
BD 10 ML Syringe Luer Lock 21G*1 inch -100 PCS BOX

BD
BD 1ml Syringe with 27G Needle | 100 Pcs Box
BD
BD Spinal Needle

BD
BD 27G Spinal Needle

BD
BD Arterial Cannula with Flow Switch - 20G | Medical Device

BD
BD Alcohol Swab - 100 Pcs Box

BD
BD Connecta 4-Way Stopcock - IV Cannula Accessories
BD
BD 10ml Syringe Discardit - 21G*1.0 inch | 100 Pcs Box