Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Explore Medicare coverage for ketamine infusions, including IV ketamine and FDA-approved esketamine (SPRAVATO). Learn about Part A, B, C, D coverage, eligibility, factors affecting costs, and what to know for treatment-resistant depression. Get clear answers on this complex topic.
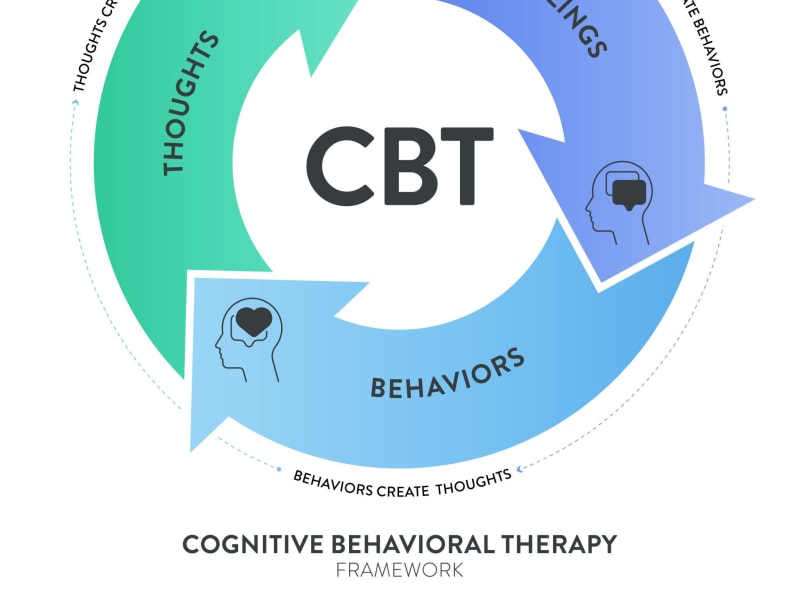
Cognitive Behavioural Therapy has one of the strongest evidence bases in mental healthcare. While professional guidance is valuable, many core CBT techniques are designed to be practised independently — and their effectiveness in self-help formats is well-documented.
April 14, 2026

Explore aversion therapy, a controversial treatment method that uses unpleasant stimuli to break unwanted habits like addiction. Learn about its methods, effectiveness, and when to seek professional help.
April 1, 2026


Ketamine, a medication primarily known as an anesthetic, has garnered significant attention in recent years for its rapid-acting antidepressant effects, particularly in individuals with treatment-resistant depression (TRD). While traditional antidepressants can take weeks to show results, ketamine infusions can provide relief within hours or days for some patients. This promising therapeutic avenue, however, often comes with a hefty price tag, leading many Medicare beneficiaries to question: Does Medicare cover ketamine infusions?
The answer, unfortunately, is not a simple yes or no. Coverage for ketamine infusions under Medicare is complex and depends on several factors, including the specific type of ketamine used, the condition being treated, the setting of administration, and the individual's Medicare plan. This comprehensive guide aims to demystify Medicare's stance on ketamine infusions, helping you understand what might be covered, what factors influence coverage decisions, and what steps you can take to explore your options.
Understanding the nuances of Medicare—including its various parts (A, B, C, D)—is crucial when seeking coverage for innovative treatments like ketamine. We'll delve into the distinctions between intravenous (IV) ketamine and its FDA-approved derivative, esketamine (SPRAVATO®) nasal spray, as their coverage pathways differ significantly. Our goal is to equip you with the knowledge needed to make informed decisions about your mental health treatment and financial planning.
Ketamine was first synthesized in 1962 and approved by the U.S. Food and Drug Administration (FDA) in 1970 as an anesthetic for medical procedures. It gained widespread use during the Vietnam War as a battlefield anesthetic due to its rapid action, analgesic properties, and minimal respiratory depression. For decades, its primary use remained in anesthesia and pain management. However, researchers began to explore its potential beyond these applications, particularly its effects on mood and cognition.
In the early 2000s, groundbreaking research revealed ketamine's profound and rapid antidepressant effects, especially in patients who had not responded to multiple conventional antidepressant treatments. This discovery sparked a revolution in mental health treatment, offering hope to millions suffering from severe and chronic depression.
Unlike conventional antidepressants that primarily target monoamine neurotransmitters like serotonin, norepinephrine, and dopamine, ketamine operates through a different mechanism. Its primary action is as an N-methyl-D-aspartate (NMDA) receptor antagonist. By blocking these receptors, ketamine triggers a cascade of events that lead to increased synaptic plasticity and the formation of new neural connections in the brain.
Specifically, ketamine is believed to:
Discover how play therapy helps children (and adults) express emotions, process trauma, and build coping skills through the natural language of play. Learn about its benefits and when to consider it.
April 1, 2026