Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Explore CAR T-cell therapy, a revolutionary immunotherapy for multiple myeloma. Learn how it works, its benefits, potential side effects, and when it's recommended.

Understanding CAR T-Cell Therapy for Multiple Myeloma Multiple myeloma is a type of blood cancer that affects plasma cells, a type of white blood cell found in the bone marrow. These abnormal plasma cells can multiply uncontrollably, crowding out healthy cells and leading to various health complications. While traditional treatments like chemotherapy, radiation, and stem cell transplants have been effective for many, some patients develop a form of the disease that becomes resistant to these therapies. For such individuals, a groundbreaking treatment called CAR T-cell therapy has emerged as a beacon of hope. What is CAR T-Cell Therapy? CAR T-cell therapy, which stands for Chimeric Antigen Receptor T-cell therapy, is a sophisticated form of immunotherapy. It's a type of gene therapy that harnesses the power of a patient's own immune system to fight cancer. Normally, T cells, a crucial component of our immune system, are responsible for identifying and destroying abnormal cells and pathogens. However, in multiple myeloma, the T cells often fail to recognize cancer cells as harmful. CAR T-cell therapy aims to correct this deficiency. How Does CAR T-Cell Therapy Work? The process begins with a procedure called leukapheresis, which is essentially a specialized blood draw. During this process, a patient's blood is passed through a machine that isolates and extracts their T cells. The remaining blood is then returned to the body. These collected T cells are then sent to a laboratory where they undergo genetic engineering. Scientists introduce special receptors, known as Chimeric Antigen Receptors (CARs), onto the surface of these T cells. These CARs are designed to specifically recognize and bind to a particular protein found on the surface of multiple myeloma cells. Once the CARs are successfully integrated, the genetically modified T cells are multiplied in the lab. This expansion process can take several weeks. After sufficient numbers of CAR T-cells are grown, they are frozen and transported back to the treatment center. Before infusion, the patient may receive a short course of chemotherapy to help reduce their existing white blood cell count, preparing their body to better receive the engineered CAR T-cells. Finally, the thawed CAR T-cells are infused back into the patient's bloodstream. These newly equipped T cells are now capable of identifying and attacking the multiple myeloma cells. Once in the body, these CAR T-cells continue to multiply, providing a sustained defense against the cancer. When is CAR T-Cell Therapy Used for Multiple Myeloma? CAR T-cell therapy is not typically a first-line treatment for multiple myeloma. It is generally considered for patients whose disease has relapsed (returned) or has become refractory (no longer responding) to other standard treatments. It represents a significant advancement for individuals who have exhausted conventional therapeutic options. While it has shown remarkable success in improving remission rates and prolonging survival, it's important to note that the cancer can still return even after successful CAR T-cell therapy. Furthermore, as this therapy was approved for multiple myeloma relatively recently (in 2017), long-term data on its efficacy and safety is still being gathered through ongoing clinical trials. What to Expect During Treatment Receiving CAR T-cell therapy involves several stages. The initial leukapheresis is usually an outpatient procedure. The subsequent laboratory phase for T-cell modification can take several weeks. The preparatory chemotherapy is typically administered a few days before the CAR T-cell infusion. The infusion itself is often an inpatient procedure, meaning you will likely need to stay at the hospital for monitoring. The early recovery phase, often referred to as the acute recovery phase, lasts for about 30 days. During this critical period, close proximity to the hospital or infusion center is essential to manage any potential complications that may arise. Potential Side Effects Like all medical treatments, CAR T-cell therapy can have side effects. Some of the most common and potentially serious side effects include: Cytokine Release Syndrome (CRS): This is a common and potentially life-threatening side effect. It occurs when the engineered T-cells release cytokines, triggering a widespread inflammatory response. Symptoms can range from mild flu-like symptoms (fever, chills, fatigue) to severe organ damage. Careful monitoring and prompt treatment are crucial for managing CRS. Neurological Toxicities: Some patients may experience neurological side effects, known as ICANS (Immune Effector Cell-Associated Neurotoxicity Syndrome). These can include confusion, difficulty speaking, tremors, seizures, and changes in consciousness. These symptoms usually appear within the first few weeks after infusion and often resolve with supportive care. Low Blood Counts: The chemotherapy used before the infusion and the CAR T-cells themselves can temporarily lower blood cell counts, increasing the risk of infection, bleeding, and anemia. Infections: Due to the weakened immune system, patients are at a higher risk of infections. Organ Damage: In severe cases, CRS and other toxicities can lead to damage in organs like the kidneys, liver, and heart. It is vital for patients to report any new or worsening symptoms to their healthcare team immediately. Recovery and Long-Term Outlook The recovery period after CAR T-cell therapy varies among individuals. The acute recovery phase requires close monitoring for about a month. Long-term recovery involves regular follow-up appointments and monitoring for up to a year to assess the treatment's effectiveness and manage any delayed side effects. While CAR T-cell therapy is often a one-time treatment, the duration of its benefit can vary. Studies have shown promising response rates, with significant percentages of patients achieving complete remission. For example, in a 2021 study, a substantial
In summary, timely diagnosis, evidence-based treatment, and prevention-focused care improve long-term health outcomes.
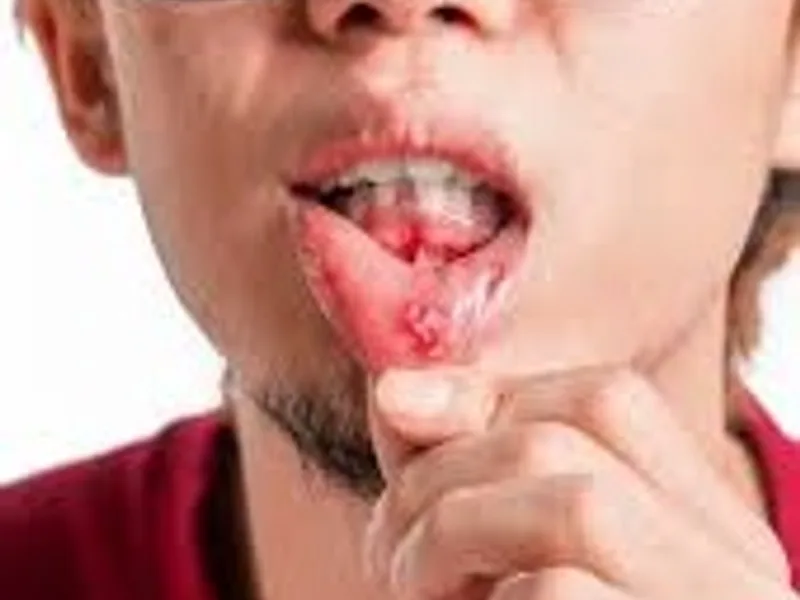
Discover 3 subtle signs of mouth cancer often dismissed as minor issues. Learn why early detection is crucial for effective treatment.
April 23, 2026

Learn about potential side effects of CML treatments like TKIs, interferon, chemotherapy, and stem cell transplants. Understand what to expect and how to communicate with your doctor for effective management.
April 1, 2026

Discover essential support resources, financial aid options, and community connections for individuals navigating life with Chronic Myeloid Leukemia (CML). Find practical advice and empathetic guidance.
April 1, 2026